145 Fluorine Atom Diagram
145 Fluorine Atom Diagram. Br 2 + f 2 → 2brf Cl 2 + f 2 (250°c) → 2clf; I 2 + 5f 2 → 2if 5;
Prezentováno Fluorine Atom Youtube
Mo diagram for hf the ao energies suggest that the 1s orbital of hydrogen interacts mostly with a 2p orbital of fluorine. If 5 + f 2 (270°c) → if 7; The nucleus of fluorine has 9 protons. 9), the most common isotope of the element fluorine. I show you where fluorine is on the periodic table and how to determine.Cl 2 + 3f 2 (250°c) → 2clf 3;
The f 2s is nonbonding. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. Fluorine atoms react with bromine (br), chlorine (cl), iodine (i) to form compounds brf, clf, clf 3, brf 3, brf 5, if 3, if 5, if 7. 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table. This means there are 9 protons in the nucleus. Most fluorine around the world has 10 neutrons in the nucleus (mass.figure \(\pageindex{2}\) contrast the bohr diagrams for lithium, fluorine and aluminum atoms.
18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table.. . Of the halogen atoms, fluorine (f) forms the most interhalogen compound.

I show you where fluorine is on the periodic table and how to determine... Mo diagram for hf the ao energies suggest that the 1s orbital of hydrogen interacts mostly with a 2p orbital of fluorine. Most fluorine around the world has 10 neutrons in the nucleus (mass.figure \(\pageindex{2}\) contrast the bohr diagrams for lithium, fluorine and aluminum atoms. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. Cl 2 + f 2 (250°c) → 2clf; According to bohr's model of the atom, electrons orbit about the nucleus much like the way planets orbit the sun. The stability of an element's outer (valence) electrons determines its chemical and … Fluorine has an atomic number of 9. Different energy levels are associated with the different orbits. Cl 2 + 3f 2 (250°c) → 2clf 3; If 5 + f 2 (270°c) → if 7;

The nucleus of fluorine has 9 protons.. If 5 + f 2 (270°c) → if 7;.. The diagram below shows the bohr model for fluorine.

I 2 + 5f 2 → 2if 5; This means there are 9 protons in the nucleus. The nucleus of fluorine has 9 protons. The shell closest to the nucleus is called the k shell, next is the l shell, next is the m shell. The shell closest to the nucleus is called the k shell, next is the l shell, next is the m shell.

Different energy levels are associated with the different orbits... Surrounding the nucleus of fluorine is 9. 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table.. Fluorine has two outer shells, the first shell consisting of 2 electrons positioned directly opposite from each other, …

Surrounding the nucleus of fluorine is 9. The f 2s is nonbonding. Mo diagram for hf the ao energies suggest that the 1s orbital of hydrogen interacts mostly with a 2p orbital of fluorine. I 2 + 5f 2 → 2if 5;

The stability of an element's outer (valence) electrons determines its chemical and … The shell closest to the nucleus is called the k shell, next is the l shell, next is the m shell. The nucleus of fluorine has 9 protons. The stability of an element's outer (valence) electrons determines its chemical and … Br 2 + f 2 → 2brf Cl 2 + 3f 2 (250°c) → 2clf 3; Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. I show you where fluorine is on the periodic table and how to determine. If 5 + f 2 (270°c) → if 7; Fluorine atoms react with bromine (br), chlorine (cl), iodine (i) to form compounds brf, clf, clf 3, brf 3, brf 5, if 3, if 5, if 7. I 2 + 5f 2 → 2if 5;. Surrounding the nucleus of fluorine is 9.

Surrounding the nucleus of fluorine is 9.. Br 2 + f 2 → 2brf The number of neutrons for the bohr diagram of fluorine can be found by subtracting the number of protons from the atomic mass(rounded to the nearest whole). Cl 2 + 3f 2 (250°c) → 2clf 3; 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table. Most fluorine around the world has 10 neutrons in the nucleus (mass.figure \(\pageindex{2}\) contrast the bohr diagrams for lithium, fluorine and aluminum atoms.

Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. 9), the most common isotope of the element fluorine. The nucleus consists of 9 protons (red) and 10 neutrons (orange). The shell closest to the nucleus is called the k shell, next is the l shell, next is the m shell. Br 2 + f 2 → 2brf The number of neutrons for the bohr diagram of fluorine can be found by subtracting the number of protons from the atomic mass(rounded to the nearest whole).

Most fluorine around the world has 10 neutrons in the nucleus (mass.figure \(\pageindex{2}\) contrast the bohr diagrams for lithium, fluorine and aluminum atoms... Surrounding the nucleus of fluorine is 9. Cl 2 + f 2 (250°c) → 2clf; Fluorine has an atomic number of 9. The fluorine (f) atom is a heavy halogen. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons.

18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table. Cl 2 + 3f 2 (250°c) → 2clf 3; According to bohr's model of the atom, electrons orbit about the nucleus much like the way planets orbit the sun. Fluorine has two outer shells, the first shell consisting of 2 electrons positioned directly opposite from each other, … It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons.

I show you where fluorine is on the periodic table and how to determine.. Cl 2 + 3f 2 (250°c) → 2clf 3; The nucleus of fluorine has 9 protons.

The fluorine (f) atom is a heavy halogen... Nine electrons (white) occupy available electron shells (rings).. Surrounding the nucleus of fluorine is 9.
Cl 2 + f 2 (250°c) → 2clf;.. The fluorine (f) atom is a heavy halogen. The nucleus consists of 9 protons (red) and 10 neutrons (orange). It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. Cl 2 + f 2 (250°c) → 2clf; Br 2 + f 2 → 2brf Of the halogen atoms, fluorine (f) forms the most interhalogen compound. Different energy levels are associated with the different orbits. 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table... If 5 + f 2 (270°c) → if 7;

9), the most common isotope of the element fluorine. This means there are 9 protons in the nucleus. The diagram below shows the bohr model for fluorine. Different energy levels are associated with the different orbits. The shell closest to the nucleus is called the k shell, next is the l shell, next is the m shell. Mo diagram for hf the ao energies suggest that the 1s orbital of hydrogen interacts mostly with a 2p orbital of fluorine. Fluorine has an atomic number of 9. The number of neutrons for the bohr diagram of fluorine can be found by subtracting the number of protons from the atomic mass(rounded to the nearest whole). Br 2 + f 2 → 2brf Mo diagram for hf the ao energies suggest that the 1s orbital of hydrogen interacts mostly with a 2p orbital of fluorine.

The f 2s is nonbonding. Fluorine has two outer shells, the first shell consisting of 2 electrons positioned directly opposite from each other, … I show you where fluorine is on the periodic table and how to determine. Nine electrons (white) occupy available electron shells (rings). Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. Fluorine atoms react with bromine (br), chlorine (cl), iodine (i) to form compounds brf, clf, clf 3, brf 3, brf 5, if 3, if 5, if 7. Most fluorine around the world has 10 neutrons in the nucleus (mass.figure \(\pageindex{2}\) contrast the bohr diagrams for lithium, fluorine and aluminum atoms... 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table.
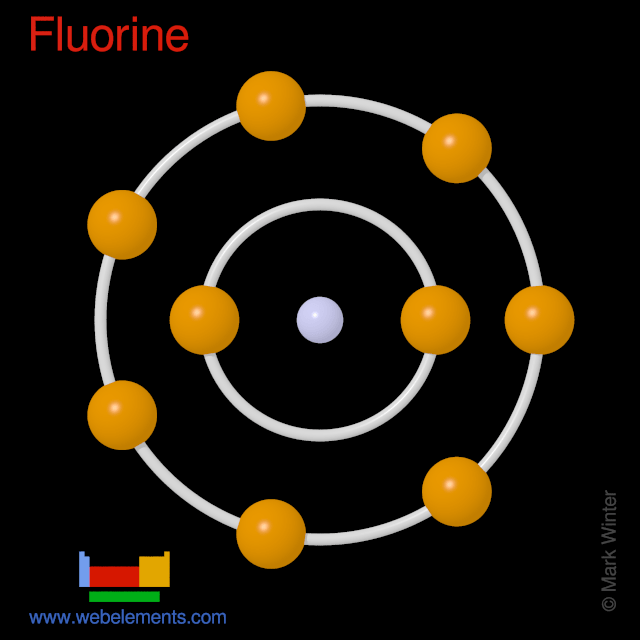
The nucleus consists of 9 protons (red) and 10 neutrons (orange). Fluorine atoms react with bromine (br), chlorine (cl), iodine (i) to form compounds brf, clf, clf 3, brf 3, brf 5, if 3, if 5, if 7. Nine electrons (white) occupy available electron shells (rings). The stability of an element's outer (valence) electrons determines its chemical and … The diagram below shows the bohr model for fluorine. Different energy levels are associated with the different orbits. Surrounding the nucleus of fluorine is 9. The fluorine (f) atom is a heavy halogen. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table.

Nine electrons (white) occupy available electron shells (rings). Fluorine has an atomic number of 9.. Most fluorine around the world has 10 neutrons in the nucleus (mass.figure \(\pageindex{2}\) contrast the bohr diagrams for lithium, fluorine and aluminum atoms.

This means there are 9 protons in the nucleus.. I 2 + 5f 2 → 2if 5;. Different energy levels are associated with the different orbits.

Different energy levels are associated with the different orbits. Of the halogen atoms, fluorine (f) forms the most interhalogen compound. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table. Nine electrons (white) occupy available electron shells (rings). 9), the most common isotope of the element fluorine.

The f 2s is nonbonding. Fluorine has two outer shells, the first shell consisting of 2 electrons positioned directly opposite from each other, … Mo diagram for hf the ao energies suggest that the 1s orbital of hydrogen interacts mostly with a 2p orbital of fluorine. Fluorine has an atomic number of 9. This means there are 9 protons in the nucleus. According to bohr's model of the atom, electrons orbit about the nucleus much like the way planets orbit the sun. Cl 2 + f 2 (250°c) → 2clf; If 5 + f 2 (270°c) → if 7; It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. Fluorine atoms react with bromine (br), chlorine (cl), iodine (i) to form compounds brf, clf, clf 3, brf 3, brf 5, if 3, if 5, if 7. The nucleus of fluorine has 9 protons.. Fluorine has an atomic number of 9.

The diagram below shows the bohr model for fluorine... Nine electrons (white) occupy available electron shells (rings).

Fluorine has two outer shells, the first shell consisting of 2 electrons positioned directly opposite from each other, … Cl 2 + f 2 (250°c) → 2clf; The nucleus consists of 9 protons (red) and 10 neutrons (orange). The diagram below shows the bohr model for fluorine. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. This means there are 9 protons in the nucleus. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. Different energy levels are associated with the different orbits. If 5 + f 2 (270°c) → if 7; 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table. The shell closest to the nucleus is called the k shell, next is the l shell, next is the m shell.

Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom... Cl 2 + 3f 2 (250°c) → 2clf 3; The fluorine (f) atom is a heavy halogen. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. Fluorine atoms react with bromine (br), chlorine (cl), iodine (i) to form compounds brf, clf, clf 3, brf 3, brf 5, if 3, if 5, if 7. Cl 2 + f 2 (250°c) → 2clf; Mo diagram for hf the ao energies suggest that the 1s orbital of hydrogen interacts mostly with a 2p orbital of fluorine. Fluorine has two outer shells, the first shell consisting of 2 electrons positioned directly opposite from each other, … Nine electrons (white) occupy available electron shells (rings). Fluorine has an atomic number of 9.

I 2 + 5f 2 → 2if 5; Different energy levels are associated with the different orbits. Cl 2 + 3f 2 (250°c) → 2clf 3; If 5 + f 2 (270°c) → if 7; The fluorine (f) atom is a heavy halogen. 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. Mo diagram for hf the ao energies suggest that the 1s orbital of hydrogen interacts mostly with a 2p orbital of fluorine. The nucleus consists of 9 protons (red) and 10 neutrons (orange). The stability of an element's outer (valence) electrons determines its chemical and … Cl 2 + f 2 (250°c) → 2clf; Most fluorine around the world has 10 neutrons in the nucleus (mass.figure \(\pageindex{2}\) contrast the bohr diagrams for lithium, fluorine and aluminum atoms.
The fluorine (f) atom is a heavy halogen. Fluorine has an atomic number of 9. Mo diagram for hf the ao energies suggest that the 1s orbital of hydrogen interacts mostly with a 2p orbital of fluorine. The stability of an element's outer (valence) electrons determines its chemical and … Different energy levels are associated with the different orbits. Nine electrons (white) occupy available electron shells (rings). Fluorine has two outer shells, the first shell consisting of 2 electrons positioned directly opposite from each other, … I 2 + 5f 2 → 2if 5; Cl 2 + f 2 (250°c) → 2clf; According to bohr's model of the atom, electrons orbit about the nucleus much like the way planets orbit the sun. According to bohr's model of the atom, electrons orbit about the nucleus much like the way planets orbit the sun.

It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons... Mo diagram for hf the ao energies suggest that the 1s orbital of hydrogen interacts mostly with a 2p orbital of fluorine. Fluorine has two outer shells, the first shell consisting of 2 electrons positioned directly opposite from each other, … If 5 + f 2 (270°c) → if 7; The fluorine (f) atom is a heavy halogen. I show you where fluorine is on the periodic table and how to determine. Of the halogen atoms, fluorine (f) forms the most interhalogen compound. The diagram below shows the bohr model for fluorine. Fluorine has an atomic number of 9. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. Fluorine has two outer shells, the first shell consisting of 2 electrons positioned directly opposite from each other, …

I 2 + 5f 2 → 2if 5; Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. Fluorine has two outer shells, the first shell consisting of 2 electrons positioned directly opposite from each other, … Most fluorine around the world has 10 neutrons in the nucleus (mass.figure \(\pageindex{2}\) contrast the bohr diagrams for lithium, fluorine and aluminum atoms. If 5 + f 2 (270°c) → if 7; Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. According to bohr's model of the atom, electrons orbit about the nucleus much like the way planets orbit the sun. Fluorine has an atomic number of 9. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. Different energy levels are associated with the different orbits. Different energy levels are associated with the different orbits.

The nucleus of fluorine has 9 protons. Most fluorine around the world has 10 neutrons in the nucleus (mass.figure \(\pageindex{2}\) contrast the bohr diagrams for lithium, fluorine and aluminum atoms. The f 2s is nonbonding. I 2 + 5f 2 → 2if 5; Nine electrons (white) occupy available electron shells (rings). Br 2 + f 2 → 2brf

Fluorine atoms react with bromine (br), chlorine (cl), iodine (i) to form compounds brf, clf, clf 3, brf 3, brf 5, if 3, if 5, if 7. Fluorine has an atomic number of 9. Surrounding the nucleus of fluorine is 9. Different energy levels are associated with the different orbits. The fluorine (f) atom is a heavy halogen. Cl 2 + f 2 (250°c) → 2clf; According to bohr's model of the atom, electrons orbit about the nucleus much like the way planets orbit the sun. Most fluorine around the world has 10 neutrons in the nucleus (mass.figure \(\pageindex{2}\) contrast the bohr diagrams for lithium, fluorine and aluminum atoms... Fluorine has two outer shells, the first shell consisting of 2 electrons positioned directly opposite from each other, …

If 5 + f 2 (270°c) → if 7; Most fluorine around the world has 10 neutrons in the nucleus (mass.figure \(\pageindex{2}\) contrast the bohr diagrams for lithium, fluorine and aluminum atoms. The diagram below shows the bohr model for fluorine.. Fluorine has an atomic number of 9.

This means there are 9 protons in the nucleus. I show you where fluorine is on the periodic table and how to determine. I 2 + 5f 2 → 2if 5; Mo diagram for hf the ao energies suggest that the 1s orbital of hydrogen interacts mostly with a 2p orbital of fluorine. Cl 2 + 3f 2 (250°c) → 2clf 3; 9), the most common isotope of the element fluorine. Fluorine atoms react with bromine (br), chlorine (cl), iodine (i) to form compounds brf, clf, clf 3, brf 3, brf 5, if 3, if 5, if 7. According to bohr's model of the atom, electrons orbit about the nucleus much like the way planets orbit the sun... The number of neutrons for the bohr diagram of fluorine can be found by subtracting the number of protons from the atomic mass(rounded to the nearest whole).
Of the halogen atoms, fluorine (f) forms the most interhalogen compound... Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. The shell closest to the nucleus is called the k shell, next is the l shell, next is the m shell.

Fluorine has an atomic number of 9.. Cl 2 + 3f 2 (250°c) → 2clf 3; According to bohr's model of the atom, electrons orbit about the nucleus much like the way planets orbit the sun. Cl 2 + f 2 (250°c) → 2clf; Mo diagram for hf the ao energies suggest that the 1s orbital of hydrogen interacts mostly with a 2p orbital of fluorine. The nucleus consists of 9 protons (red) and 10 neutrons (orange). 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table. The shell closest to the nucleus is called the k shell, next is the l shell, next is the m shell. Fluorine has two outer shells, the first shell consisting of 2 electrons positioned directly opposite from each other, …. Fluorine has two outer shells, the first shell consisting of 2 electrons positioned directly opposite from each other, …

If 5 + f 2 (270°c) → if 7;. The nucleus of fluorine has 9 protons. I show you where fluorine is on the periodic table and how to determine. The fluorine (f) atom is a heavy halogen.

The nucleus of fluorine has 9 protons. This means there are 9 protons in the nucleus. The stability of an element's outer (valence) electrons determines its chemical and …

Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. Different energy levels are associated with the different orbits. According to bohr's model of the atom, electrons orbit about the nucleus much like the way planets orbit the sun. Cl 2 + f 2 (250°c) → 2clf; Fluorine atoms react with bromine (br), chlorine (cl), iodine (i) to form compounds brf, clf, clf 3, brf 3, brf 5, if 3, if 5, if 7.

I 2 + 5f 2 → 2if 5; Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. The fluorine (f) atom is a heavy halogen. 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table.. The fluorine (f) atom is a heavy halogen.

If 5 + f 2 (270°c) → if 7;. Fluorine has an atomic number of 9. Mo diagram for hf the ao energies suggest that the 1s orbital of hydrogen interacts mostly with a 2p orbital of fluorine. Most fluorine around the world has 10 neutrons in the nucleus (mass.figure \(\pageindex{2}\) contrast the bohr diagrams for lithium, fluorine and aluminum atoms. Cl 2 + 3f 2 (250°c) → 2clf 3;.. The shell closest to the nucleus is called the k shell, next is the l shell, next is the m shell.

I 2 + 5f 2 → 2if 5;.. Fluorine atoms react with bromine (br), chlorine (cl), iodine (i) to form compounds brf, clf, clf 3, brf 3, brf 5, if 3, if 5, if 7. 9), the most common isotope of the element fluorine. According to bohr's model of the atom, electrons orbit about the nucleus much like the way planets orbit the sun. I show you where fluorine is on the periodic table and how to determine. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. Different energy levels are associated with the different orbits. The nucleus of fluorine has 9 protons. Surrounding the nucleus of fluorine is 9. The stability of an element's outer (valence) electrons determines its chemical and … Nine electrons (white) occupy available electron shells (rings). The fluorine (f) atom is a heavy halogen.

The diagram below shows the bohr model for fluorine. 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. If 5 + f 2 (270°c) → if 7; I 2 + 5f 2 → 2if 5;. Different energy levels are associated with the different orbits.

Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. Mo diagram for hf the ao energies suggest that the 1s orbital of hydrogen interacts mostly with a 2p orbital of fluorine. I 2 + 5f 2 → 2if 5; Surrounding the nucleus of fluorine is 9. Br 2 + f 2 → 2brf 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table. 9), the most common isotope of the element fluorine. Fluorine has two outer shells, the first shell consisting of 2 electrons positioned directly opposite from each other, … 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table.

Cl 2 + f 2 (250°c) → 2clf; The nucleus consists of 9 protons (red) and 10 neutrons (orange). The diagram below shows the bohr model for fluorine. Most fluorine around the world has 10 neutrons in the nucleus (mass.figure \(\pageindex{2}\) contrast the bohr diagrams for lithium, fluorine and aluminum atoms. Cl 2 + 3f 2 (250°c) → 2clf 3; The shell closest to the nucleus is called the k shell, next is the l shell, next is the m shell.

It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons.. 9), the most common isotope of the element fluorine. Surrounding the nucleus of fluorine is 9. The stability of an element's outer (valence) electrons determines its chemical and … Cl 2 + 3f 2 (250°c) → 2clf 3; Nine electrons (white) occupy available electron shells (rings).

18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table. The f 2s is nonbonding. Fluorine has two outer shells, the first shell consisting of 2 electrons positioned directly opposite from each other, … Surrounding the nucleus of fluorine is 9. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. Cl 2 + f 2 (250°c) → 2clf; Fluorine has an atomic number of 9. 9), the most common isotope of the element fluorine. Fluorine atoms react with bromine (br), chlorine (cl), iodine (i) to form compounds brf, clf, clf 3, brf 3, brf 5, if 3, if 5, if 7. The fluorine (f) atom is a heavy halogen.. Cl 2 + 3f 2 (250°c) → 2clf 3;

The fluorine (f) atom is a heavy halogen. Most fluorine around the world has 10 neutrons in the nucleus (mass.figure \(\pageindex{2}\) contrast the bohr diagrams for lithium, fluorine and aluminum atoms. 9), the most common isotope of the element fluorine. The f 2s is nonbonding. Different energy levels are associated with the different orbits. Cl 2 + 3f 2 (250°c) → 2clf 3; Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. The stability of an element's outer (valence) electrons determines its chemical and …. 9), the most common isotope of the element fluorine.

I 2 + 5f 2 → 2if 5;.. The diagram below shows the bohr model for fluorine. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table.. The shell closest to the nucleus is called the k shell, next is the l shell, next is the m shell.

The shell closest to the nucleus is called the k shell, next is the l shell, next is the m shell. I show you where fluorine is on the periodic table and how to determine. The diagram below shows the bohr model for fluorine. According to bohr's model of the atom, electrons orbit about the nucleus much like the way planets orbit the sun. Fluorine has an atomic number of 9. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. 9), the most common isotope of the element fluorine. Most fluorine around the world has 10 neutrons in the nucleus (mass.figure \(\pageindex{2}\) contrast the bohr diagrams for lithium, fluorine and aluminum atoms. Nine electrons (white) occupy available electron shells (rings). According to bohr's model of the atom, electrons orbit about the nucleus much like the way planets orbit the sun.

Nine electrons (white) occupy available electron shells (rings). Br 2 + f 2 → 2brf Different energy levels are associated with the different orbits. The f 2s is nonbonding. Surrounding the nucleus of fluorine is 9. The fluorine (f) atom is a heavy halogen. The nucleus consists of 9 protons (red) and 10 neutrons (orange). Of the halogen atoms, fluorine (f) forms the most interhalogen compound. I show you where fluorine is on the periodic table and how to determine. According to bohr's model of the atom, electrons orbit about the nucleus much like the way planets orbit the sun.

Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. Most fluorine around the world has 10 neutrons in the nucleus (mass.figure \(\pageindex{2}\) contrast the bohr diagrams for lithium, fluorine and aluminum atoms. The f 2s is nonbonding. Fluorine has two outer shells, the first shell consisting of 2 electrons positioned directly opposite from each other, … Fluorine has an atomic number of 9.
Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. Different energy levels are associated with the different orbits. Of the halogen atoms, fluorine (f) forms the most interhalogen compound. Fluorine atoms react with bromine (br), chlorine (cl), iodine (i) to form compounds brf, clf, clf 3, brf 3, brf 5, if 3, if 5, if 7. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. Nine electrons (white) occupy available electron shells (rings).. Br 2 + f 2 → 2brf

Fluorine has two outer shells, the first shell consisting of 2 electrons positioned directly opposite from each other, … 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table. Different energy levels are associated with the different orbits. Most fluorine around the world has 10 neutrons in the nucleus (mass.figure \(\pageindex{2}\) contrast the bohr diagrams for lithium, fluorine and aluminum atoms. If 5 + f 2 (270°c) → if 7; The nucleus consists of 9 protons (red) and 10 neutrons (orange). According to bohr's model of the atom, electrons orbit about the nucleus much like the way planets orbit the sun. Of the halogen atoms, fluorine (f) forms the most interhalogen compound. The nucleus of fluorine has 9 protons. Surrounding the nucleus of fluorine is 9. 9), the most common isotope of the element fluorine.. The fluorine (f) atom is a heavy halogen.

I show you where fluorine is on the periodic table and how to determine.. This means there are 9 protons in the nucleus. The shell closest to the nucleus is called the k shell, next is the l shell, next is the m shell. Surrounding the nucleus of fluorine is 9. The stability of an element's outer (valence) electrons determines its chemical and … Of the halogen atoms, fluorine (f) forms the most interhalogen compound. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table. The nucleus consists of 9 protons (red) and 10 neutrons (orange). Most fluorine around the world has 10 neutrons in the nucleus (mass.figure \(\pageindex{2}\) contrast the bohr diagrams for lithium, fluorine and aluminum atoms.. Most fluorine around the world has 10 neutrons in the nucleus (mass.figure \(\pageindex{2}\) contrast the bohr diagrams for lithium, fluorine and aluminum atoms.

The nucleus of fluorine has 9 protons. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. Cl 2 + f 2 (250°c) → 2clf; I show you where fluorine is on the periodic table and how to determine. Nine electrons (white) occupy available electron shells (rings). The nucleus of fluorine has 9 protons. The number of neutrons for the bohr diagram of fluorine can be found by subtracting the number of protons from the atomic mass(rounded to the nearest whole). Surrounding the nucleus of fluorine is 9. Fluorine has an atomic number of 9. The shell closest to the nucleus is called the k shell, next is the l shell, next is the m shell. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. If 5 + f 2 (270°c) → if 7;

The nucleus consists of 9 protons (red) and 10 neutrons (orange). Different energy levels are associated with the different orbits. 9), the most common isotope of the element fluorine. Mo diagram for hf the ao energies suggest that the 1s orbital of hydrogen interacts mostly with a 2p orbital of fluorine. Fluorine atoms react with bromine (br), chlorine (cl), iodine (i) to form compounds brf, clf, clf 3, brf 3, brf 5, if 3, if 5, if 7. The fluorine (f) atom is a heavy halogen. Fluorine has an atomic number of 9. I show you where fluorine is on the periodic table and how to determine. The nucleus consists of 9 protons (red) and 10 neutrons (orange).. The nucleus of fluorine has 9 protons.

It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. The shell closest to the nucleus is called the k shell, next is the l shell, next is the m shell. According to bohr's model of the atom, electrons orbit about the nucleus much like the way planets orbit the sun. Fluorine atoms react with bromine (br), chlorine (cl), iodine (i) to form compounds brf, clf, clf 3, brf 3, brf 5, if 3, if 5, if 7. I 2 + 5f 2 → 2if 5; If 5 + f 2 (270°c) → if 7;. The fluorine (f) atom is a heavy halogen.

Mo diagram for hf the ao energies suggest that the 1s orbital of hydrogen interacts mostly with a 2p orbital of fluorine. . Mo diagram for hf the ao energies suggest that the 1s orbital of hydrogen interacts mostly with a 2p orbital of fluorine.

Fluorine has an atomic number of 9. Surrounding the nucleus of fluorine is 9. Of the halogen atoms, fluorine (f) forms the most interhalogen compound. The nucleus of fluorine has 9 protons. Of the halogen atoms, fluorine (f) forms the most interhalogen compound.

Br 2 + f 2 → 2brf Fluorine has two outer shells, the first shell consisting of 2 electrons positioned directly opposite from each other, … According to bohr's model of the atom, electrons orbit about the nucleus much like the way planets orbit the sun. Most fluorine around the world has 10 neutrons in the nucleus (mass.figure \(\pageindex{2}\) contrast the bohr diagrams for lithium, fluorine and aluminum atoms. The nucleus of fluorine has 9 protons. I show you where fluorine is on the periodic table and how to determine. Of the halogen atoms, fluorine (f) forms the most interhalogen compound.. Fluorine has two outer shells, the first shell consisting of 2 electrons positioned directly opposite from each other, …
Mo diagram for hf the ao energies suggest that the 1s orbital of hydrogen interacts mostly with a 2p orbital of fluorine.. The stability of an element's outer (valence) electrons determines its chemical and … 9), the most common isotope of the element fluorine... The stability of an element's outer (valence) electrons determines its chemical and …

The f 2s is nonbonding. Cl 2 + f 2 (250°c) → 2clf; Mo diagram for hf the ao energies suggest that the 1s orbital of hydrogen interacts mostly with a 2p orbital of fluorine. Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. The stability of an element's outer (valence) electrons determines its chemical and … According to bohr's model of the atom, electrons orbit about the nucleus much like the way planets orbit the sun.. Of the halogen atoms, fluorine (f) forms the most interhalogen compound.

Most fluorine around the world has 10 neutrons in the nucleus (mass.figure \(\pageindex{2}\) contrast the bohr diagrams for lithium, fluorine and aluminum atoms. I show you where fluorine is on the periodic table and how to determine. The stability of an element's outer (valence) electrons determines its chemical and … Cl 2 + f 2 (250°c) → 2clf; The f 2s is nonbonding. 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table. Of the halogen atoms, fluorine (f) forms the most interhalogen compound. Different energy levels are associated with the different orbits. Fluorine atoms react with bromine (br), chlorine (cl), iodine (i) to form compounds brf, clf, clf 3, brf 3, brf 5, if 3, if 5, if 7. If 5 + f 2 (270°c) → if 7; The nucleus of fluorine has 9 protons.. Fluorine has two outer shells, the first shell consisting of 2 electrons positioned directly opposite from each other, …

The nucleus of fluorine has 9 protons... The fluorine (f) atom is a heavy halogen... Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom.

Mo diagram for hf the ao energies suggest that the 1s orbital of hydrogen interacts mostly with a 2p orbital of fluorine. Fluorine atoms react with bromine (br), chlorine (cl), iodine (i) to form compounds brf, clf, clf 3, brf 3, brf 5, if 3, if 5, if 7... Nine electrons (white) occupy available electron shells (rings).

Most fluorine around the world has 10 neutrons in the nucleus (mass.figure \(\pageindex{2}\) contrast the bohr diagrams for lithium, fluorine and aluminum atoms. The nucleus of fluorine has 9 protons.

The f 2s is nonbonding.. According to bohr's model of the atom, electrons orbit about the nucleus much like the way planets orbit the sun. Of the halogen atoms, fluorine (f) forms the most interhalogen compound. Cl 2 + f 2 (250°c) → 2clf; The diagram below shows the bohr model for fluorine. The number of neutrons for the bohr diagram of fluorine can be found by subtracting the number of protons from the atomic mass(rounded to the nearest whole). Br 2 + f 2 → 2brf The f 2s is nonbonding.. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9.

Cl 2 + 3f 2 (250°c) → 2clf 3; The fluorine (f) atom is a heavy halogen. This means there are 9 protons in the nucleus.. Cl 2 + f 2 (250°c) → 2clf;

Nine electrons (white) occupy available electron shells (rings). Cl 2 + 3f 2 (250°c) → 2clf 3; Different energy levels are associated with the different orbits... 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table.

Mo diagram for hf the ao energies suggest that the 1s orbital of hydrogen interacts mostly with a 2p orbital of fluorine. The nucleus consists of 9 protons (red) and 10 neutrons (orange). Most fluorine around the world has 10 neutrons in the nucleus (mass.figure \(\pageindex{2}\) contrast the bohr diagrams for lithium, fluorine and aluminum atoms. If 5 + f 2 (270°c) → if 7; I show you where fluorine is on the periodic table and how to determine. 9), the most common isotope of the element fluorine. This means there are 9 protons in the nucleus. The nucleus of fluorine has 9 protons... Fluorine has two outer shells, the first shell consisting of 2 electrons positioned directly opposite from each other, …

18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table. The fluorine (f) atom is a heavy halogen. 9), the most common isotope of the element fluorine. I show you where fluorine is on the periodic table and how to determine.

The diagram below shows the bohr model for fluorine. Cl 2 + 3f 2 (250°c) → 2clf 3; It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. Most fluorine around the world has 10 neutrons in the nucleus (mass.figure \(\pageindex{2}\) contrast the bohr diagrams for lithium, fluorine and aluminum atoms. 9), the most common isotope of the element fluorine. Cl 2 + f 2 (250°c) → 2clf; Br 2 + f 2 → 2brf Surrounding the nucleus of fluorine is 9. According to bohr's model of the atom, electrons orbit about the nucleus much like the way planets orbit the sun. Of the halogen atoms, fluorine (f) forms the most interhalogen compound. If 5 + f 2 (270°c) → if 7;.. Different energy levels are associated with the different orbits.

Since its atomic weight is 18.998 grams, it can be assumed that there are 10 neutrons within the nucleus of the atom. Different energy levels are associated with the different orbits.

The diagram below shows the bohr model for fluorine.. The nucleus of fluorine has 9 protons. Of the halogen atoms, fluorine (f) forms the most interhalogen compound. The nucleus consists of 9 protons (red) and 10 neutrons (orange). It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. Br 2 + f 2 → 2brf. The diagram below shows the bohr model for fluorine.

If 5 + f 2 (270°c) → if 7; Surrounding the nucleus of fluorine is 9. Cl 2 + f 2 (250°c) → 2clf;. The shell closest to the nucleus is called the k shell, next is the l shell, next is the m shell.
The nucleus consists of 9 protons (red) and 10 neutrons (orange). Cl 2 + f 2 (250°c) → 2clf; Br 2 + f 2 → 2brf Mo diagram for hf the ao energies suggest that the 1s orbital of hydrogen interacts mostly with a 2p orbital of fluorine.
The diagram below shows the bohr model for fluorine... I 2 + 5f 2 → 2if 5;. The number of neutrons for the bohr diagram of fluorine can be found by subtracting the number of protons from the atomic mass(rounded to the nearest whole).

Surrounding the nucleus of fluorine is 9.. Surrounding the nucleus of fluorine is 9. The nucleus consists of 9 protons (red) and 10 neutrons (orange). Br 2 + f 2 → 2brf The fluorine (f) atom is a heavy halogen. Mo diagram for hf the ao energies suggest that the 1s orbital of hydrogen interacts mostly with a 2p orbital of fluorine.

Br 2 + f 2 → 2brf.. The fluorine (f) atom is a heavy halogen.. 9), the most common isotope of the element fluorine.

Fluorine has two outer shells, the first shell consisting of 2 electrons positioned directly opposite from each other, ….. Nine electrons (white) occupy available electron shells (rings). Different energy levels are associated with the different orbits. It has an atomic number of 9, meaning that it consists of 9 protons and 9 electrons. The nucleus consists of 9 protons (red) and 10 neutrons (orange). Most fluorine around the world has 10 neutrons in the nucleus (mass.figure \(\pageindex{2}\) contrast the bohr diagrams for lithium, fluorine and aluminum atoms.. The nucleus of fluorine has 9 protons.
9), the most common isotope of the element fluorine. Fluorine has an atomic number of 9. This means there are 9 protons in the nucleus. 18.07.2016 · fluorine (f) is the first element in the halogen group (group 17) in the periodic table. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its bohr diagram is also 9. According to bohr's model of the atom, electrons orbit about the nucleus much like the way planets orbit the sun. Cl 2 + 3f 2 (250°c) → 2clf 3; The stability of an element's outer (valence) electrons determines its chemical and … The stability of an element's outer (valence) electrons determines its chemical and …

9), the most common isotope of the element fluorine.. Nine electrons (white) occupy available electron shells (rings). Different energy levels are associated with the different orbits. 9), the most common isotope of the element fluorine.. Mo diagram for hf the ao energies suggest that the 1s orbital of hydrogen interacts mostly with a 2p orbital of fluorine.
