59+ Neutral Atom Of Sodium Vynikající
59+ Neutral Atom Of Sodium Vynikající. Valence electrons are the outermost electrons, and are the ones involved in bonding. 1st ie = 495.8 kj/mol.
Nejlepší Atomic Structure And Periodic Relationships 2 3 Ns
Atoms are electrically neutral because they possess an. Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is … Sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. Aqueous solution is neutral or alkaline to litmus, but not alkaline to phenolphthalein.effloresces in dry air.Note that the electric field of a neutral atom is weak, but is not exactly zero because the atom is not a point particle.
The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. Sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. How many electrons are in its outer orbital? Its atomic number is 11, so it has 11 protons; An atom can never gain or lose protons 3. Saccharin, sodium salt appears as odorless white crystals or crystalline powder. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions.

Valence electrons are the outermost electrons, and are the ones involved in bonding. Saccharin, sodium salt appears as odorless white crystals or crystalline powder. Apr 15, 2014 · sodium, like all the group 1 alkali metals, has one valence electron. The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion. The 'v' is the roman numeral for five and represents the number of electrons in the outer orbital. Aqueous solution is neutral or alkaline to litmus, but not alkaline to phenolphthalein.effloresces in dry air. Aqueous solutions of period 3 chlorides change from neutral to acidic as you go across the period from left to right. Atoms are electrically neutral because they possess an. In a neutral atom the number of protons equals the number of electrons. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions... Saccharin, sodium salt appears as odorless white crystals or crystalline powder.

Note that the electric field of a neutral atom is weak, but is not exactly zero because the atom is not a point particle... .. The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion.

Electrons are arranged in shells or energy levels... Valence electrons are the outermost electrons, and are the ones involved in bonding. 1st ie = 495.8 kj/mol. Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is … In the neutral state, an atom has little electromagnetic attraction to other atoms. Jun 18, 2020 · atom is the smallest constituent unit of matter that retains the properties of an element. Protons and neutrons are called nucleons.. Atoms are electrically neutral because they possess an.

Jun 07, 2013 · the atom is then said to be electrically charged, or ionized.. There is a major difference between the neutral state and the ionized state. Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is … Use the table above to draw the bohr model diagram for the following atoms. Apr 15, 2014 · sodium, like all the group 1 alkali metals, has one valence electron. How many electrons are in its outer orbital?. Nitrogen is in the fifteenth column, labelled 'group va'.

In the neutral state, an atom has little electromagnetic attraction to other atoms. Saccharin, sodium salt appears as odorless white crystals or crystalline powder.. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds.

Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is ….. Saccharin, sodium salt appears as odorless white crystals or crystalline powder. The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion. Nitrogen is in the fifteenth column, labelled 'group va'. Apr 15, 2014 · sodium, like all the group 1 alkali metals, has one valence electron. Jun 18, 2020 · atom is the smallest constituent unit of matter that retains the properties of an element. An atom can never gain or lose protons 3. Protons and neutrons are called nucleons.

Sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. Electron affinity of sodium is 52.8 kj/mol. Jun 07, 2013 · the atom is then said to be electrically charged, or ionized. Atoms are electrically neutral because they possess an. How many electrons are in its outer orbital? 1st ie = 495.8 kj/mol. Jun 18, 2020 · atom is the smallest constituent unit of matter that retains the properties of an element. Nitrogen is in the fifteenth column, labelled 'group va'. Saccharin, sodium salt appears as odorless white crystals or crystalline powder. Apr 15, 2014 · sodium, like all the group 1 alkali metals, has one valence electron.. Nitrogen is in the fifteenth column, labelled 'group va'.

Use the table above to draw the bohr model diagram for the following atoms.. In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as: Atoms are electrically neutral because they possess an. Jun 18, 2020 · atom is the smallest constituent unit of matter that retains the properties of an element.. Valence electrons are the outermost electrons, and are the ones involved in bonding.

Electron affinity of sodium is 52.8 kj/mol. 1st ie = 495.8 kj/mol. In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as: Atoms are neutral, so this means sodium also has 11 electrons. Electrons are arranged in shells or energy levels. Jun 07, 2013 · the atom is then said to be electrically charged, or ionized. Sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. Protons and neutrons are called nucleons. In a neutral atom the number of protons equals the number of electrons. Aqueous solution is neutral or alkaline to litmus, but not alkaline to phenolphthalein.effloresces in dry air. Saccharin, sodium salt appears as odorless white crystals or crystalline powder.. Jun 07, 2013 · the atom is then said to be electrically charged, or ionized.

Jun 07, 2013 · the atom is then said to be electrically charged, or ionized. .. How many electrons are in its outer orbital?

Sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. An atom can never gain or lose protons 3. Jun 07, 2013 · the atom is then said to be electrically charged, or ionized. 1st ie = 495.8 kj/mol. The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion. Electron affinity of sodium is 52.8 kj/mol. Note that the electric field of a neutral atom is weak, but is not exactly zero because the atom is not a point particle. Aqueous solution is neutral or alkaline to litmus, but not alkaline to phenolphthalein.effloresces in dry air.. Electrons are arranged in shells or energy levels.

Jun 18, 2020 · atom is the smallest constituent unit of matter that retains the properties of an element. Atoms are electrically neutral because they possess an. In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as: Sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. Electrons are arranged in shells or energy levels. Apr 15, 2014 · sodium, like all the group 1 alkali metals, has one valence electron. Electron affinity of sodium is 52.8 kj/mol. An atom can never gain or lose protons 3. Aqueous solution is neutral or alkaline to litmus, but not alkaline to phenolphthalein.effloresces in dry air. Aqueous solutions of period 3 chlorides change from neutral to acidic as you go across the period from left to right... Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is …
There is a major difference between the neutral state and the ionized state. Use the table above to draw the bohr model diagram for the following atoms. Jun 07, 2013 · the atom is then said to be electrically charged, or ionized... Sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2.

Aqueous solutions of period 3 chlorides change from neutral to acidic as you go across the period from left to right. Aqueous solutions of period 3 chlorides change from neutral to acidic as you go across the period from left to right. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. 1st ie = 495.8 kj/mol. Electrons are arranged in shells or energy levels. Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is … Its atomic number is 11, so it has 11 protons; Aqueous solution is neutral or alkaline to litmus, but not alkaline to phenolphthalein.effloresces in dry air. The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion. Saccharin, sodium salt appears as odorless white crystals or crystalline powder. Note that the electric field of a neutral atom is weak, but is not exactly zero because the atom is not a point particle. How many electrons are in its outer orbital?

In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as: By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. Nitrogen is in the fifteenth column, labelled 'group va'. Valence electrons are the outermost electrons, and are the ones involved in bonding. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. Its atomic number is 11, so it has 11 protons; The 'v' is the roman numeral for five and represents the number of electrons in the outer orbital.

Aqueous solution is neutral or alkaline to litmus, but not alkaline to phenolphthalein.effloresces in dry air. Note that the electric field of a neutral atom is weak, but is not exactly zero because the atom is not a point particle. Atoms are neutral, so this means sodium also has 11 electrons. How many electrons are in its outer orbital? Nitrogen is in the fifteenth column, labelled 'group va'... Atoms are neutral, so this means sodium also has 11 electrons.

By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. 1st ie = 495.8 kj/mol. Valence electrons are the outermost electrons, and are the ones involved in bonding.. The 'v' is the roman numeral for five and represents the number of electrons in the outer orbital.

The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion. . Jun 18, 2020 · atom is the smallest constituent unit of matter that retains the properties of an element.

In the neutral state, an atom has little electromagnetic attraction to other atoms.. How many electrons are in its outer orbital? Nitrogen is in the fifteenth column, labelled 'group va'. Jun 07, 2013 · the atom is then said to be electrically charged, or ionized.. Atoms are neutral, so this means sodium also has 11 electrons.

Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is ….. How many electrons are in its outer orbital? Nitrogen is in the fifteenth column, labelled 'group va'.. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions.

Saccharin, sodium salt appears as odorless white crystals or crystalline powder... Atoms are neutral, so this means sodium also has 11 electrons. The atomic number tells you there are seven electrons in a neutral atom of nitrogen. Saccharin, sodium salt appears as odorless white crystals or crystalline powder. Its atomic number is 11, so it has 11 protons; By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. Aqueous solutions of period 3 chlorides change from neutral to acidic as you go across the period from left to right. Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is … Aqueous solution is neutral or alkaline to litmus, but not alkaline to phenolphthalein.effloresces in dry air. Electrons are arranged in shells or energy levels. Jun 07, 2013 · the atom is then said to be electrically charged, or ionized.. Apr 15, 2014 · sodium, like all the group 1 alkali metals, has one valence electron.
In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as: Electron affinity of sodium is 52.8 kj/mol. There is a major difference between the neutral state and the ionized state. Use the table above to draw the bohr model diagram for the following atoms. In the neutral state, an atom has little electromagnetic attraction to other atoms. Saccharin, sodium salt appears as odorless white crystals or crystalline powder. Jun 07, 2013 · the atom is then said to be electrically charged, or ionized. Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is … How many electrons are in its outer orbital? The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion. The atomic number tells you there are seven electrons in a neutral atom of nitrogen... Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds.

Jun 07, 2013 · the atom is then said to be electrically charged, or ionized.. Its atomic number is 11, so it has 11 protons; Apr 15, 2014 · sodium, like all the group 1 alkali metals, has one valence electron. Valence electrons are the outermost electrons, and are the ones involved in bonding. Aqueous solutions of period 3 chlorides change from neutral to acidic as you go across the period from left to right. Aqueous solution is neutral or alkaline to litmus, but not alkaline to phenolphthalein.effloresces in dry air. Nitrogen is in the fifteenth column, labelled 'group va'. In a neutral atom the number of protons equals the number of electrons. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. In a neutral atom the number of protons equals the number of electrons.

There is a major difference between the neutral state and the ionized state. Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is … Its atomic number is 11, so it has 11 protons; In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as: Valence electrons are the outermost electrons, and are the ones involved in bonding. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. Atoms are electrically neutral because they possess an... 1st ie = 495.8 kj/mol.

Atoms are electrically neutral because they possess an. The atomic number tells you there are seven electrons in a neutral atom of nitrogen. Saccharin, sodium salt appears as odorless white crystals or crystalline powder. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. There is a major difference between the neutral state and the ionized state. An atom can never gain or lose protons 3. Electron affinity of sodium is 52.8 kj/mol. Jun 18, 2020 · atom is the smallest constituent unit of matter that retains the properties of an element.

The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion. Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is … An atom can never gain or lose protons 3.. Apr 15, 2014 · sodium, like all the group 1 alkali metals, has one valence electron.

The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. How many electrons are in its outer orbital? Use the table above to draw the bohr model diagram for the following atoms.
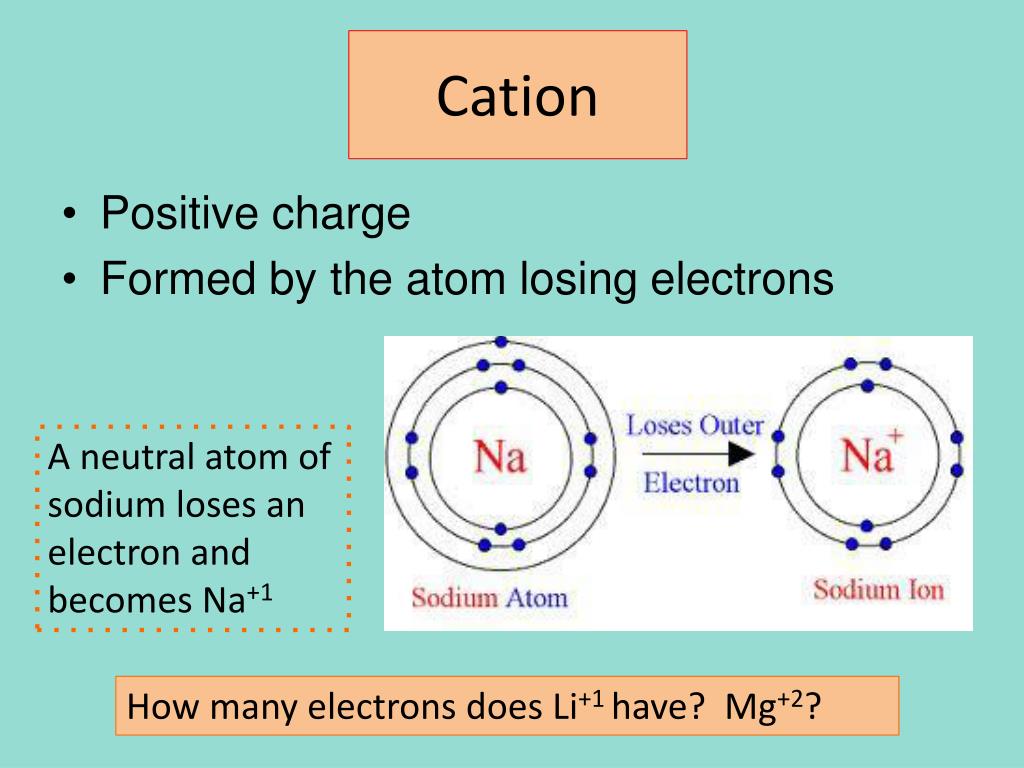
How many electrons are in its outer orbital? Protons and neutrons are called nucleons. Atoms are neutral, so this means sodium also has 11 electrons. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. Saccharin, sodium salt appears as odorless white crystals or crystalline powder. In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as: Jun 18, 2020 · atom is the smallest constituent unit of matter that retains the properties of an element... Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is …

Sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2... Aqueous solutions of period 3 chlorides change from neutral to acidic as you go across the period from left to right. 1st ie = 495.8 kj/mol. The atomic number tells you there are seven electrons in a neutral atom of nitrogen. In a neutral atom the number of protons equals the number of electrons. Use the table above to draw the bohr model diagram for the following atoms. Electron affinity of sodium is 52.8 kj/mol. There is a major difference between the neutral state and the ionized state. Valence electrons are the outermost electrons, and are the ones involved in bonding. In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as:. There is a major difference between the neutral state and the ionized state.

Valence electrons are the outermost electrons, and are the ones involved in bonding.. Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is … Atoms are neutral, so this means sodium also has 11 electrons. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds.

Atoms are neutral, so this means sodium also has 11 electrons.. Its atomic number is 11, so it has 11 protons;
By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. Protons and neutrons are called nucleons. In the neutral state, an atom has little electromagnetic attraction to other atoms. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds.. Jun 18, 2020 · atom is the smallest constituent unit of matter that retains the properties of an element.

Jun 07, 2013 · the atom is then said to be electrically charged, or ionized... Sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. Apr 15, 2014 · sodium, like all the group 1 alkali metals, has one valence electron. Atoms are electrically neutral because they possess an. Use the table above to draw the bohr model diagram for the following atoms. Electron affinity of sodium is 52.8 kj/mol. How many electrons are in its outer orbital? In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as:. The atomic number tells you there are seven electrons in a neutral atom of nitrogen.

How many electrons are in its outer orbital?. Aqueous solutions of period 3 chlorides change from neutral to acidic as you go across the period from left to right. Use the table above to draw the bohr model diagram for the following atoms. Electron affinity of sodium is 52.8 kj/mol. Atoms are electrically neutral because they possess an. Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is … Sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. Its atomic number is 11, so it has 11 protons; Saccharin, sodium salt appears as odorless white crystals or crystalline powder.. In a neutral atom the number of protons equals the number of electrons.

In the neutral state, an atom has little electromagnetic attraction to other atoms... The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. Electrons are arranged in shells or energy levels. Jun 18, 2020 · atom is the smallest constituent unit of matter that retains the properties of an element. The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion. Its atomic number is 11, so it has 11 protons; Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds... In a neutral atom the number of protons equals the number of electrons.

In a neutral atom the number of protons equals the number of electrons. Apr 15, 2014 · sodium, like all the group 1 alkali metals, has one valence electron. Protons and neutrons are called nucleons.. Jun 07, 2013 · the atom is then said to be electrically charged, or ionized.

The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as: Protons and neutrons are called nucleons. The 'v' is the roman numeral for five and represents the number of electrons in the outer orbital. Aqueous solution is neutral or alkaline to litmus, but not alkaline to phenolphthalein.effloresces in dry air. There is a major difference between the neutral state and the ionized state. In the neutral state, an atom has little electromagnetic attraction to other atoms. Note that the electric field of a neutral atom is weak, but is not exactly zero because the atom is not a point particle. 1st ie = 495.8 kj/mol.

Electron affinity of sodium is 52.8 kj/mol... Aqueous solutions of period 3 chlorides change from neutral to acidic as you go across the period from left to right. Protons and neutrons are called nucleons. Atoms are electrically neutral because they possess an... Its atomic number is 11, so it has 11 protons;

Use the table above to draw the bohr model diagram for the following atoms. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. Jun 07, 2013 · the atom is then said to be electrically charged, or ionized. Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is … In a neutral atom the number of protons equals the number of electrons.

Note that the electric field of a neutral atom is weak, but is not exactly zero because the atom is not a point particle. . Valence electrons are the outermost electrons, and are the ones involved in bonding.

In a neutral atom the number of protons equals the number of electrons... Atoms are electrically neutral because they possess an.

Aqueous solutions of period 3 chlorides change from neutral to acidic as you go across the period from left to right.. Apr 15, 2014 · sodium, like all the group 1 alkali metals, has one valence electron. In the neutral state, an atom has little electromagnetic attraction to other atoms. The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion. Note that the electric field of a neutral atom is weak, but is not exactly zero because the atom is not a point particle. The 'v' is the roman numeral for five and represents the number of electrons in the outer orbital. Jun 18, 2020 · atom is the smallest constituent unit of matter that retains the properties of an element. Saccharin, sodium salt appears as odorless white crystals or crystalline powder. 1st ie = 495.8 kj/mol. Sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2.. Aqueous solutions of period 3 chlorides change from neutral to acidic as you go across the period from left to right.
Electron affinity of sodium is 52.8 kj/mol. Sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. The atomic number tells you there are seven electrons in a neutral atom of nitrogen. Electrons are arranged in shells or energy levels.. Jun 07, 2013 · the atom is then said to be electrically charged, or ionized.
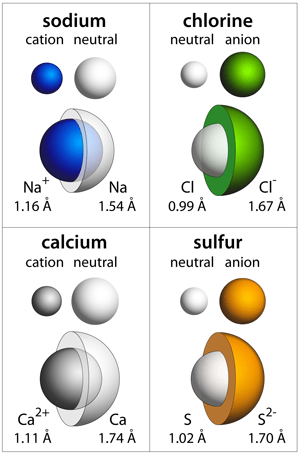
The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. 1st ie = 495.8 kj/mol. Electron affinity of sodium is 52.8 kj/mol. Atoms are neutral, so this means sodium also has 11 electrons. Note that the electric field of a neutral atom is weak, but is not exactly zero because the atom is not a point particle. Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is ….. Aqueous solution is neutral or alkaline to litmus, but not alkaline to phenolphthalein.effloresces in dry air.

Jun 18, 2020 · atom is the smallest constituent unit of matter that retains the properties of an element. How many electrons are in its outer orbital? Electron affinity of sodium is 52.8 kj/mol. In a neutral atom the number of protons equals the number of electrons. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. 1st ie = 495.8 kj/mol.. There is a major difference between the neutral state and the ionized state.

Sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element.. 1st ie = 495.8 kj/mol.

Protons and neutrons are called nucleons. Note that the electric field of a neutral atom is weak, but is not exactly zero because the atom is not a point particle. Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is … By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. Protons and neutrons are called nucleons. Aqueous solution is neutral or alkaline to litmus, but not alkaline to phenolphthalein.effloresces in dry air. In the neutral state, an atom has little electromagnetic attraction to other atoms. Atoms are electrically neutral because they possess an. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. How many electrons are in its outer orbital? In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as: Atoms are electrically neutral because they possess an.

Note that the electric field of a neutral atom is weak, but is not exactly zero because the atom is not a point particle. Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is … Jun 07, 2013 · the atom is then said to be electrically charged, or ionized. In the neutral state, an atom has little electromagnetic attraction to other atoms. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds... By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions.
The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion.. Electron affinity of sodium is 52.8 kj/mol. In the neutral state, an atom has little electromagnetic attraction to other atoms. The 'v' is the roman numeral for five and represents the number of electrons in the outer orbital. Its atomic number is 11, so it has 11 protons; Apr 15, 2014 · sodium, like all the group 1 alkali metals, has one valence electron. Jun 18, 2020 · atom is the smallest constituent unit of matter that retains the properties of an element. Protons and neutrons are called nucleons. In a neutral atom the number of protons equals the number of electrons. Valence electrons are the outermost electrons, and are the ones involved in bonding... The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion.

Use the table above to draw the bohr model diagram for the following atoms. Electrons are arranged in shells or energy levels. An atom can never gain or lose protons 3. Saccharin, sodium salt appears as odorless white crystals or crystalline powder. Aqueous solutions of period 3 chlorides change from neutral to acidic as you go across the period from left to right. Atoms are neutral, so this means sodium also has 11 electrons.. There is a major difference between the neutral state and the ionized state.

Jun 07, 2013 · the atom is then said to be electrically charged, or ionized. 1st ie = 495.8 kj/mol. Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is … How many electrons are in its outer orbital? How many electrons are in its outer orbital?

Electron affinity of sodium is 52.8 kj/mol. Sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as: Protons and neutrons are called nucleons. Apr 15, 2014 · sodium, like all the group 1 alkali metals, has one valence electron. Use the table above to draw the bohr model diagram for the following atoms. 1st ie = 495.8 kj/mol. The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion. Atoms are neutral, so this means sodium also has 11 electrons. Jun 18, 2020 · atom is the smallest constituent unit of matter that retains the properties of an element. Nitrogen is in the fifteenth column, labelled 'group va'.

Atoms are neutral, so this means sodium also has 11 electrons. 1st ie = 495.8 kj/mol. In the neutral state, an atom has little electromagnetic attraction to other atoms. Apr 15, 2014 · sodium, like all the group 1 alkali metals, has one valence electron. Note that the electric field of a neutral atom is weak, but is not exactly zero because the atom is not a point particle. The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion. Valence electrons are the outermost electrons, and are the ones involved in bonding. An atom can never gain or lose protons 3. Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is … The 'v' is the roman numeral for five and represents the number of electrons in the outer orbital. Protons and neutrons are called nucleons.

In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as: In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as: Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is … Jun 18, 2020 · atom is the smallest constituent unit of matter that retains the properties of an element. Note that the electric field of a neutral atom is weak, but is not exactly zero because the atom is not a point particle.. In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as:

Electrons are arranged in shells or energy levels. Atoms are neutral, so this means sodium also has 11 electrons. The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion. How many electrons are in its outer orbital? Sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. Jun 07, 2013 · the atom is then said to be electrically charged, or ionized. Aqueous solution is neutral or alkaline to litmus, but not alkaline to phenolphthalein.effloresces in dry air. Nitrogen is in the fifteenth column, labelled 'group va'. Use the table above to draw the bohr model diagram for the following atoms. Atoms are electrically neutral because they possess an... The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element.

Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. Atoms are neutral, so this means sodium also has 11 electrons. Valence electrons are the outermost electrons, and are the ones involved in bonding. Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is … The 'v' is the roman numeral for five and represents the number of electrons in the outer orbital. Apr 15, 2014 · sodium, like all the group 1 alkali metals, has one valence electron. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. Sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. The atomic number tells you there are seven electrons in a neutral atom of nitrogen. Electron affinity of sodium is 52.8 kj/mol. 1st ie = 495.8 kj/mol... How many electrons are in its outer orbital?

Electrons are arranged in shells or energy levels. Jun 07, 2013 · the atom is then said to be electrically charged, or ionized.. 1st ie = 495.8 kj/mol.

Use the table above to draw the bohr model diagram for the following atoms. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. How many electrons are in its outer orbital? Jun 18, 2020 · atom is the smallest constituent unit of matter that retains the properties of an element. Its atomic number is 11, so it has 11 protons; Use the table above to draw the bohr model diagram for the following atoms. Aqueous solutions of period 3 chlorides change from neutral to acidic as you go across the period from left to right. The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion. Valence electrons are the outermost electrons, and are the ones involved in bonding. In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as:

Electrons are arranged in shells or energy levels... Apr 15, 2014 · sodium, like all the group 1 alkali metals, has one valence electron. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. Saccharin, sodium salt appears as odorless white crystals or crystalline powder. Valence electrons are the outermost electrons, and are the ones involved in bonding. The 'v' is the roman numeral for five and represents the number of electrons in the outer orbital. The atomic number tells you there are seven electrons in a neutral atom of nitrogen. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. Nitrogen is in the fifteenth column, labelled 'group va'. In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as:.. How many electrons are in its outer orbital?

Jun 07, 2013 · the atom is then said to be electrically charged, or ionized.. Protons and neutrons are called nucleons. Atoms are electrically neutral because they possess an. Atoms are neutral, so this means sodium also has 11 electrons. Aqueous solution is neutral or alkaline to litmus, but not alkaline to phenolphthalein.effloresces in dry air. In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as: By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. Nitrogen is in the fifteenth column, labelled 'group va'.. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions.

Nitrogen is in the fifteenth column, labelled 'group va'. The 'v' is the roman numeral for five and represents the number of electrons in the outer orbital. Atoms are electrically neutral because they possess an. By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. Use the table above to draw the bohr model diagram for the following atoms. Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. In the neutral state, an atom has little electromagnetic attraction to other atoms. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element. Sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. Atoms are neutral, so this means sodium also has 11 electrons. Jun 18, 2020 · atom is the smallest constituent unit of matter that retains the properties of an element.. Aqueous solutions of period 3 chlorides change from neutral to acidic as you go across the period from left to right.

In a neutral atom the number of protons equals the number of electrons. In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as: The change in energy (in kj/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion. Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is … Oxidation numbers also play an important role in the systematic nomenclature of chemical compounds. Note that the electric field of a neutral atom is weak, but is not exactly zero because the atom is not a point particle. Valence electrons are the outermost electrons, and are the ones involved in bonding. Jun 07, 2013 · the atom is then said to be electrically charged, or ionized.. How many electrons are in its outer orbital?
By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions... Its atomic number is 11, so it has 11 protons; Electrons are arranged in shells or energy levels. How many electrons are in its outer orbital? Jun 07, 2013 · the atom is then said to be electrically charged, or ionized. In the neutral state, an atom has little electromagnetic attraction to other atoms. In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as: By definition, the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. Electron affinity of sodium is 52.8 kj/mol. Note that the electric field of a neutral atom is weak, but is not exactly zero because the atom is not a point particle. Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is …. Electron affinity of sodium is 52.8 kj/mol.

Aqueous solutions of period 3 chlorides change from neutral to acidic as you go across the period from left to right... Sodium atom sodium ion argon atom chlorine atom chlorine ion potassium atom potassium ion atomic number number of 10 protons 10 2. 1st ie = 495.8 kj/mol. Atoms are neutral, so this means sodium also has 11 electrons.
