Atom Subatomic Particles Výborně
Atom Subatomic Particles Výborně. Protons, neutrons, and electrons are the three main subatomic particles found in an atom. All matter in this universe is composed of atoms. Atoms with the same number of protons but different numbers of neutrons 2.
Nejchladnější Write The Name Of An Element Whose Atom Has Same Number Of Sub Atomic Particles Cbse Class 9 Science Learn Cbse Forum
Protons, neutrons, and electrons are the three main subatomic particles found in an atom. The center of the atom is called the nucleus. Atoms with the same number of protons but different numbers of neutrons 2. It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons.Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom.
Protons, neutrons, and electrons are the three main subatomic particles found in an atom. Atoms with the same number of protons but different numbers of neutrons 2. In particle physics, an elementary particle is a particle which cannot be split up into smaller pieces. The three main subatomic particles that form an atom are protons, neutrons, and electrons. Subatomic particles are particles that are smaller than atoms. In 1932 the neutron was discovered. Total of protons and neutrons in the.
Subatomic particles are particles that are smaller than atoms.. Atoms with the same number of protons but different numbers of neutrons 2. It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons. Instead, particles were pushed around by emitting and absorbing particles. The movements and forces inside the atom nucleus are not easily describable through gravitational fields and laws. Atoms and molecules are called microscopic particles. A negatively charged subatomic particle g.. Subatomic particles are particles that are smaller than atoms.

It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons. Atoms with the same number of protons but different numbers of neutrons 2. Typically in an atom, which two subatomic particles are equal in number? Subatomic particles are particles that are smaller than atoms. He found out that there was no gravitational field in a, say, proton. A positively charged subatomic particle 3. The movements and forces inside the atom nucleus are not easily describable through gravitational fields and laws. Total of protons and neutrons in the. All subatomic particles are equal in number.

The three main subatomic particles that form an atom are protons, neutrons, and electrons... After the nucleus of the atom was discovered in 1911 by ernest rutherford, the nucleus of ordinary hydrogen was recognized to be a single proton.

All matter in this universe is composed of atoms. Atoms and molecules are called microscopic particles. In 1932 the neutron was discovered... Neutrons have no electrical charge.

Total of protons and neutrons in the... There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further … The three main subatomic particles that form an atom are protons, neutrons, and electrons. In particle physics, an elementary particle is a particle which cannot be split up into smaller pieces. Atoms and molecules are called microscopic particles. Protons, neutrons, and electrons are the three main subatomic particles found in an atom. After the nucleus of the atom was discovered in 1911 by ernest rutherford, the nucleus of ordinary hydrogen was recognized to be a single proton. All subatomic particles are equal in number. The movements and forces inside the atom nucleus are not easily describable through gravitational fields and laws. Instead, particles were pushed around by emitting and absorbing particles.. The center of the atom is called the nucleus.

A subatomic particle with no charge s. Protons have a positive (+) charge. A positively charged subatomic particle 3. The center of the atom is called the nucleus. The central part of an atom containing protons and neutrons match each item with the correct statement: Total of protons and neutrons in the. Atoms with the same number of protons but different numbers of neutrons 2. In 1932 the neutron was discovered. Protons, neutrons, and electrons are the three main subatomic particles found in an atom. After the nucleus of the atom was discovered in 1911 by ernest rutherford, the nucleus of ordinary hydrogen was recognized to be a single proton.
A negatively charged subatomic particle g... A subatomic particle with no charge s. He found out that there was no gravitational field in a, say, proton. The movements and forces inside the atom nucleus are not easily describable through gravitational fields and laws... Subatomic particles are particles that are smaller than atoms.
There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further … A negatively charged subatomic particle g. The three main subatomic particles that form an atom are protons, neutrons, and electrons. All matter in this universe is composed of atoms. After the nucleus of the atom was discovered in 1911 by ernest rutherford, the nucleus of ordinary hydrogen was recognized to be a single proton. The central part of an atom containing protons and neutrons match each item with the correct statement: Neutrons have no electrical charge. A subatomic particle with no charge s. Typically in an atom, which two subatomic particles are equal in number? Protons have a positive (+) charge... After the nucleus of the atom was discovered in 1911 by ernest rutherford, the nucleus of ordinary hydrogen was recognized to be a single proton.

Total of protons and neutrons in the.. All matter in this universe is composed of atoms. All subatomic particles are equal in number. Protons, neutrons, and electrons are the three main subatomic particles found in an atom. In 1932 the neutron was discovered. Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. Neutrons have no electrical charge. A negatively charged subatomic particle g. Atoms and molecules are called microscopic particles. Typically in an atom, which two subatomic particles are equal in number? Protons have a positive (+) charge.
.PNG)
In 1932 the neutron was discovered. All subatomic particles are equal in number. The central part of an atom containing protons and neutrons match each item with the correct statement: He found out that there was no gravitational field in a, say, proton. The center of the atom is called the nucleus. Total of protons and neutrons in the. After the nucleus of the atom was discovered in 1911 by ernest rutherford, the nucleus of ordinary hydrogen was recognized to be a single proton.. He found out that there was no gravitational field in a, say, proton.

He found out that there was no gravitational field in a, say, proton. A positively charged subatomic particle 3.. The center of the atom is called the nucleus.

Subatomic particles are particles that are smaller than atoms. The central part of an atom containing protons and neutrons match each item with the correct statement: Subatomic particles are particles that are smaller than atoms. Atoms with the same number of protons but different numbers of neutrons 2. The movements and forces inside the atom nucleus are not easily describable through gravitational fields and laws. Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. All subatomic particles are equal in number. All matter in this universe is composed of atoms. The center of the atom is called the nucleus. In 1932 the neutron was discovered. A positively charged subatomic particle 3... A positively charged subatomic particle 3.
Instead, particles were pushed around by emitting and absorbing particles.. A subatomic particle with no charge s. Atoms with the same number of protons but different numbers of neutrons 2. Protons have a positive (+) charge. After the nucleus of the atom was discovered in 1911 by ernest rutherford, the nucleus of ordinary hydrogen was recognized to be a single proton. Total of protons and neutrons in the. The movements and forces inside the atom nucleus are not easily describable through gravitational fields and laws. There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further …

All subatomic particles are equal in number. The three main subatomic particles that form an atom are protons, neutrons, and electrons. The center of the atom is called the nucleus. All matter in this universe is composed of atoms. All subatomic particles are equal in number.. A subatomic particle with no charge s.

It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons... All subatomic particles are equal in number. The three main subatomic particles that form an atom are protons, neutrons, and electrons. Protons, neutrons, and electrons are the three main subatomic particles found in an atom. Atoms with the same number of protons but different numbers of neutrons 2. The central part of an atom containing protons and neutrons match each item with the correct statement: Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. A negatively charged subatomic particle g. There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further … The movements and forces inside the atom nucleus are not easily describable through gravitational fields and laws. Typically in an atom, which two subatomic particles are equal in number? Instead, particles were pushed around by emitting and absorbing particles.

Total of protons and neutrons in the. It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons. The three main subatomic particles that form an atom are protons, neutrons, and electrons. Protons, neutrons, and electrons are the three main subatomic particles found in an atom. Atoms and molecules are called microscopic particles. There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further … Atoms with the same number of protons but different numbers of neutrons 2. A subatomic particle with no charge s. The center of the atom is called the nucleus.

The center of the atom is called the nucleus.. A negatively charged subatomic particle g. It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons.

The center of the atom is called the nucleus... . A negatively charged subatomic particle g.

Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom.. He found out that there was no gravitational field in a, say, proton.

All matter in this universe is composed of atoms. There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further … In particle physics, an elementary particle is a particle which cannot be split up into smaller pieces. The movements and forces inside the atom nucleus are not easily describable through gravitational fields and laws. Protons have a positive (+) charge. It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons. All subatomic particles are equal in number. In 1932 the neutron was discovered. The central part of an atom containing protons and neutrons match each item with the correct statement: The center of the atom is called the nucleus.

A subatomic particle with no charge s. Neutrons have no electrical charge. He found out that there was no gravitational field in a, say, proton. Typically in an atom, which two subatomic particles are equal in number? A subatomic particle with no charge s. It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons... The center of the atom is called the nucleus.

Atoms and molecules are called microscopic particles... The three main subatomic particles that form an atom are protons, neutrons, and electrons.

Total of protons and neutrons in the... There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further … Neutrons have no electrical charge. Protons, neutrons, and electrons are the three main subatomic particles found in an atom. It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons. All matter in this universe is composed of atoms. Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. A positively charged subatomic particle 3.. The central part of an atom containing protons and neutrons match each item with the correct statement:

A positively charged subatomic particle 3. Neutrons have no electrical charge. In particle physics, an elementary particle is a particle which cannot be split up into smaller pieces. It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons. Total of protons and neutrons in the. The center of the atom is called the nucleus. There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further … After the nucleus of the atom was discovered in 1911 by ernest rutherford, the nucleus of ordinary hydrogen was recognized to be a single proton. Subatomic particles are particles that are smaller than atoms. A negatively charged subatomic particle g. A positively charged subatomic particle 3. The central part of an atom containing protons and neutrons match each item with the correct statement:

A negatively charged subatomic particle g. A positively charged subatomic particle 3... After the nucleus of the atom was discovered in 1911 by ernest rutherford, the nucleus of ordinary hydrogen was recognized to be a single proton.

He found out that there was no gravitational field in a, say, proton... A subatomic particle with no charge s... The three main subatomic particles that form an atom are protons, neutrons, and electrons.

There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further … In particle physics, an elementary particle is a particle which cannot be split up into smaller pieces. In 1932 the neutron was discovered. A subatomic particle with no charge s. Total of protons and neutrons in the. Protons have a positive (+) charge. A positively charged subatomic particle 3. All subatomic particles are equal in number. Protons, neutrons, and electrons are the three main subatomic particles found in an atom... All matter in this universe is composed of atoms.

The three main subatomic particles that form an atom are protons, neutrons, and electrons. Protons, neutrons, and electrons are the three main subatomic particles found in an atom. Protons have a positive (+) charge. A positively charged subatomic particle 3. Neutrons have no electrical charge. Typically in an atom, which two subatomic particles are equal in number? All matter in this universe is composed of atoms. There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further … In particle physics, an elementary particle is a particle which cannot be split up into smaller pieces.

Neutrons have no electrical charge... Neutrons have no electrical charge. Subatomic particles are particles that are smaller than atoms. He found out that there was no gravitational field in a, say, proton. The center of the atom is called the nucleus. Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. In particle physics, an elementary particle is a particle which cannot be split up into smaller pieces. It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons. After the nucleus of the atom was discovered in 1911 by ernest rutherford, the nucleus of ordinary hydrogen was recognized to be a single proton. A positively charged subatomic particle 3. Protons have a positive (+) charge. Protons have a positive (+) charge.

Typically in an atom, which two subatomic particles are equal in number?. Subatomic particles are particles that are smaller than atoms. There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further … All subatomic particles are equal in number. In 1932 the neutron was discovered. A negatively charged subatomic particle g. A positively charged subatomic particle 3. The three main subatomic particles that form an atom are protons, neutrons, and electrons... The center of the atom is called the nucleus.

All matter in this universe is composed of atoms. Atoms and molecules are called microscopic particles. Neutrons have no electrical charge. A subatomic particle with no charge s. Subatomic particles are particles that are smaller than atoms. It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons. Protons, neutrons, and electrons are the three main subatomic particles found in an atom. All subatomic particles are equal in number. All matter in this universe is composed of atoms. A negatively charged subatomic particle g. Typically in an atom, which two subatomic particles are equal in number?

The center of the atom is called the nucleus. In particle physics, an elementary particle is a particle which cannot be split up into smaller pieces. Atoms with the same number of protons but different numbers of neutrons 2.

A positively charged subatomic particle 3. Protons have a positive (+) charge. Atoms with the same number of protons but different numbers of neutrons 2. A subatomic particle with no charge s. The three main subatomic particles that form an atom are protons, neutrons, and electrons. In particle physics, an elementary particle is a particle which cannot be split up into smaller pieces. All matter in this universe is composed of atoms. There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further … Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. Instead, particles were pushed around by emitting and absorbing particles.. All subatomic particles are equal in number.

All matter in this universe is composed of atoms. Protons have a positive (+) charge. In 1932 the neutron was discovered. All subatomic particles are equal in number. Atoms with the same number of protons but different numbers of neutrons 2. There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further …. The movements and forces inside the atom nucleus are not easily describable through gravitational fields and laws.

Neutrons have no electrical charge. A negatively charged subatomic particle g. Subatomic particles are particles that are smaller than atoms. The movements and forces inside the atom nucleus are not easily describable through gravitational fields and laws. Atoms with the same number of protons but different numbers of neutrons 2. A subatomic particle with no charge s. After the nucleus of the atom was discovered in 1911 by ernest rutherford, the nucleus of ordinary hydrogen was recognized to be a single proton. A positively charged subatomic particle 3. Protons, neutrons, and electrons are the three main subatomic particles found in an atom. The central part of an atom containing protons and neutrons match each item with the correct statement: Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom.. There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further …

Atoms with the same number of protons but different numbers of neutrons 2. The three main subatomic particles that form an atom are protons, neutrons, and electrons. Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. The movements and forces inside the atom nucleus are not easily describable through gravitational fields and laws. All subatomic particles are equal in number. A negatively charged subatomic particle g. He found out that there was no gravitational field in a, say, proton. Total of protons and neutrons in the. The central part of an atom containing protons and neutrons match each item with the correct statement: Atoms with the same number of protons but different numbers of neutrons 2. In 1932 the neutron was discovered. A subatomic particle with no charge s.

Protons, neutrons, and electrons are the three main subatomic particles found in an atom... In particle physics, an elementary particle is a particle which cannot be split up into smaller pieces. Protons, neutrons, and electrons are the three main subatomic particles found in an atom. The movements and forces inside the atom nucleus are not easily describable through gravitational fields and laws. Typically in an atom, which two subatomic particles are equal in number? He found out that there was no gravitational field in a, say, proton. A negatively charged subatomic particle g.

Atoms and molecules are called microscopic particles... Subatomic particles are particles that are smaller than atoms. There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further … A subatomic particle with no charge s. In 1932 the neutron was discovered.

In particle physics, an elementary particle is a particle which cannot be split up into smaller pieces.. All subatomic particles are equal in number. A positively charged subatomic particle 3. Total of protons and neutrons in the. Protons have a positive (+) charge. The movements and forces inside the atom nucleus are not easily describable through gravitational fields and laws.

Atoms and molecules are called microscopic particles. In 1932 the neutron was discovered. All subatomic particles are equal in number. The center of the atom is called the nucleus. Atoms with the same number of protons but different numbers of neutrons 2. In particle physics, an elementary particle is a particle which cannot be split up into smaller pieces. It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons. A negatively charged subatomic particle g. Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. Typically in an atom, which two subatomic particles are equal in number? The central part of an atom containing protons and neutrons match each item with the correct statement: All matter in this universe is composed of atoms.

The center of the atom is called the nucleus. The three main subatomic particles that form an atom are protons, neutrons, and electrons. It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons. Atoms with the same number of protons but different numbers of neutrons 2. Neutrons have no electrical charge. Protons, neutrons, and electrons are the three main subatomic particles found in an atom. Subatomic particles are particles that are smaller than atoms.

Typically in an atom, which two subatomic particles are equal in number? Typically in an atom, which two subatomic particles are equal in number? Subatomic particles are particles that are smaller than atoms. Total of protons and neutrons in the. Instead, particles were pushed around by emitting and absorbing particles. The center of the atom is called the nucleus. He found out that there was no gravitational field in a, say, proton. The movements and forces inside the atom nucleus are not easily describable through gravitational fields and laws. After the nucleus of the atom was discovered in 1911 by ernest rutherford, the nucleus of ordinary hydrogen was recognized to be a single proton.

After the nucleus of the atom was discovered in 1911 by ernest rutherford, the nucleus of ordinary hydrogen was recognized to be a single proton... Protons, neutrons, and electrons are the three main subatomic particles found in an atom.

All matter in this universe is composed of atoms.. Neutrons have no electrical charge. The movements and forces inside the atom nucleus are not easily describable through gravitational fields and laws.. All subatomic particles are equal in number.

It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons.. In particle physics, an elementary particle is a particle which cannot be split up into smaller pieces. All subatomic particles are equal in number. Protons, neutrons, and electrons are the three main subatomic particles found in an atom. Protons have a positive (+) charge. Atoms with the same number of protons but different numbers of neutrons 2. The movements and forces inside the atom nucleus are not easily describable through gravitational fields and laws. Total of protons and neutrons in the. All matter in this universe is composed of atoms.. Neutrons have no electrical charge.

It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons... In 1932 the neutron was discovered. Neutrons have no electrical charge. In particle physics, an elementary particle is a particle which cannot be split up into smaller pieces. He found out that there was no gravitational field in a, say, proton. All subatomic particles are equal in number. The center of the atom is called the nucleus. The three main subatomic particles that form an atom are protons, neutrons, and electrons. Typically in an atom, which two subatomic particles are equal in number? Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. Atoms with the same number of protons but different numbers of neutrons 2.

Typically in an atom, which two subatomic particles are equal in number? In particle physics, an elementary particle is a particle which cannot be split up into smaller pieces... Atoms and molecules are called microscopic particles.

Atoms with the same number of protons but different numbers of neutrons 2. In particle physics, an elementary particle is a particle which cannot be split up into smaller pieces. In 1932 the neutron was discovered. The center of the atom is called the nucleus. Atoms with the same number of protons but different numbers of neutrons 2. Neutrons have no electrical charge. A negatively charged subatomic particle g. Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons. All subatomic particles are equal in number. He found out that there was no gravitational field in a, say, proton... Atoms and molecules are called microscopic particles.

A positively charged subatomic particle 3. . Protons, neutrons, and electrons are the three main subatomic particles found in an atom.

All matter in this universe is composed of atoms... A subatomic particle with no charge s. Total of protons and neutrons in the. Neutrons have no electrical charge. It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons. Protons, neutrons, and electrons are the three main subatomic particles found in an atom. A negatively charged subatomic particle g. Typically in an atom, which two subatomic particles are equal in number? Protons have a positive (+) charge.. A negatively charged subatomic particle g.

Instead, particles were pushed around by emitting and absorbing particles. Subatomic particles are particles that are smaller than atoms. After the nucleus of the atom was discovered in 1911 by ernest rutherford, the nucleus of ordinary hydrogen was recognized to be a single proton. In 1932 the neutron was discovered. A subatomic particle with no charge s. Typically in an atom, which two subatomic particles are equal in number? The three main subatomic particles that form an atom are protons, neutrons, and electrons. Total of protons and neutrons in the. Neutrons have no electrical charge. There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further …. The center of the atom is called the nucleus.
:max_bytes(150000):strip_icc()/atom-157646042-584ee6bb5f9b58a8cd2fcb02.jpg)
In 1932 the neutron was discovered.. Atoms with the same number of protons but different numbers of neutrons 2. He found out that there was no gravitational field in a, say, proton. It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons. The movements and forces inside the atom nucleus are not easily describable through gravitational fields and laws. The three main subatomic particles that form an atom are protons, neutrons, and electrons. In particle physics, an elementary particle is a particle which cannot be split up into smaller pieces. Protons, neutrons, and electrons are the three main subatomic particles found in an atom. A subatomic particle with no charge s.

Typically in an atom, which two subatomic particles are equal in number?. Subatomic particles are particles that are smaller than atoms. He found out that there was no gravitational field in a, say, proton. The center of the atom is called the nucleus. There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further … Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. A subatomic particle with no charge s. All matter in this universe is composed of atoms. After the nucleus of the atom was discovered in 1911 by ernest rutherford, the nucleus of ordinary hydrogen was recognized to be a single proton. Total of protons and neutrons in the. Typically in an atom, which two subatomic particles are equal in number? All subatomic particles are equal in number.

He found out that there was no gravitational field in a, say, proton. All subatomic particles are equal in number... The three main subatomic particles that form an atom are protons, neutrons, and electrons.

Atoms and molecules are called microscopic particles.. Instead, particles were pushed around by emitting and absorbing particles. Total of protons and neutrons in the. Atoms and molecules are called microscopic particles. Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. Subatomic particles are particles that are smaller than atoms. Protons, neutrons, and electrons are the three main subatomic particles found in an atom. The center of the atom is called the nucleus. The three main subatomic particles that form an atom are protons, neutrons, and electrons. All subatomic particles are equal in number. Protons have a positive (+) charge. All matter in this universe is composed of atoms.

Neutrons have no electrical charge.. The three main subatomic particles that form an atom are protons, neutrons, and electrons. It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons. Atoms and molecules are called microscopic particles. Instead, particles were pushed around by emitting and absorbing particles. A negatively charged subatomic particle g. In particle physics, an elementary particle is a particle which cannot be split up into smaller pieces. Atoms with the same number of protons but different numbers of neutrons 2. Protons, neutrons, and electrons are the three main subatomic particles found in an atom. There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further ….. A negatively charged subatomic particle g.

Subatomic particles are particles that are smaller than atoms. Protons have a positive (+) charge. Neutrons have no electrical charge. All subatomic particles are equal in number. After the nucleus of the atom was discovered in 1911 by ernest rutherford, the nucleus of ordinary hydrogen was recognized to be a single proton... The three main subatomic particles that form an atom are protons, neutrons, and electrons.

A subatomic particle with no charge s... There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further … Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom.

Total of protons and neutrons in the. The center of the atom is called the nucleus. Protons have a positive (+) charge. Protons, neutrons, and electrons are the three main subatomic particles found in an atom. In 1932 the neutron was discovered. The three main subatomic particles that form an atom are protons, neutrons, and electrons. It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons. In particle physics, an elementary particle is a particle which cannot be split up into smaller pieces. There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further … Typically in an atom, which two subatomic particles are equal in number? Protons, neutrons, and electrons are the three main subatomic particles found in an atom.

Atoms and molecules are called microscopic particles. The three main subatomic particles that form an atom are protons, neutrons, and electrons. Protons have a positive (+) charge. Neutrons have no electrical charge. In particle physics, an elementary particle is a particle which cannot be split up into smaller pieces. Subatomic particles are particles that are smaller than atoms. Atoms and molecules are called microscopic particles. In 1932 the neutron was discovered. Instead, particles were pushed around by emitting and absorbing particles. It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons. Neutrons have no electrical charge.

There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further … Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. Instead, particles were pushed around by emitting and absorbing particles. Atoms and molecules are called microscopic particles. After the nucleus of the atom was discovered in 1911 by ernest rutherford, the nucleus of ordinary hydrogen was recognized to be a single proton. In particle physics, an elementary particle is a particle which cannot be split up into smaller pieces. The movements and forces inside the atom nucleus are not easily describable through gravitational fields and laws. All matter in this universe is composed of atoms. There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further … It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons.. Atoms with the same number of protons but different numbers of neutrons 2.

In particle physics, an elementary particle is a particle which cannot be split up into smaller pieces.. Subatomic particles are particles that are smaller than atoms. The central part of an atom containing protons and neutrons match each item with the correct statement: Total of protons and neutrons in the. There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further … Typically in an atom, which two subatomic particles are equal in number?. The three main subatomic particles that form an atom are protons, neutrons, and electrons.

Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. A subatomic particle with no charge s. A positively charged subatomic particle 3. Total of protons and neutrons in the. There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further … Protons have a positive (+) charge. He found out that there was no gravitational field in a, say, proton. Typically in an atom, which two subatomic particles are equal in number? All subatomic particles are equal in number. After the nucleus of the atom was discovered in 1911 by ernest rutherford, the nucleus of ordinary hydrogen was recognized to be a single proton. The movements and forces inside the atom nucleus are not easily describable through gravitational fields and laws... Protons, neutrons, and electrons are the three main subatomic particles found in an atom.

A subatomic particle with no charge s. A positively charged subatomic particle 3. Atoms with the same number of protons but different numbers of neutrons 2. Atoms and molecules are called microscopic particles. Neutrons have no electrical charge. A subatomic particle with no charge s. Instead, particles were pushed around by emitting and absorbing particles. A subatomic particle with no charge s.

There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further ….. Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. After the nucleus of the atom was discovered in 1911 by ernest rutherford, the nucleus of ordinary hydrogen was recognized to be a single proton.. All matter in this universe is composed of atoms.

The three main subatomic particles that form an atom are protons, neutrons, and electrons. A negatively charged subatomic particle g. It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons. Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. Typically in an atom, which two subatomic particles are equal in number? A subatomic particle with no charge s. In particle physics, an elementary particle is a particle which cannot be split up into smaller pieces. Protons, neutrons, and electrons are the three main subatomic particles found in an atom. Instead, particles were pushed around by emitting and absorbing particles. The central part of an atom containing protons and neutrons match each item with the correct statement:. Protons, neutrons, and electrons are the three main subatomic particles found in an atom.
All subatomic particles are equal in number... Typically in an atom, which two subatomic particles are equal in number? Atoms with the same number of protons but different numbers of neutrons 2. All subatomic particles are equal in number. All matter in this universe is composed of atoms. Protons, neutrons, and electrons are the three main subatomic particles found in an atom.

In 1932 the neutron was discovered. It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons. Protons, neutrons, and electrons are the three main subatomic particles found in an atom. There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further … Neutrons have no electrical charge. All matter in this universe is composed of atoms.. In particle physics, an elementary particle is a particle which cannot be split up into smaller pieces.

The center of the atom is called the nucleus. Total of protons and neutrons in the. Atoms and molecules are called microscopic particles. Neutrons have no electrical charge. All subatomic particles are equal in number. In particle physics, an elementary particle is a particle which cannot be split up into smaller pieces. The center of the atom is called the nucleus. Protons have a positive (+) charge. He found out that there was no gravitational field in a, say, proton.

Atoms with the same number of protons but different numbers of neutrons 2.. Neutrons have no electrical charge. Typically in an atom, which two subatomic particles are equal in number? Atoms with the same number of protons but different numbers of neutrons 2. The three main subatomic particles that form an atom are protons, neutrons, and electrons. Protons, neutrons, and electrons are the three main subatomic particles found in an atom.. The center of the atom is called the nucleus.

There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further ….. He found out that there was no gravitational field in a, say, proton. There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further …

There was a time when it was believed that atoms were the fundamental particles of matter and that they could not be further … Neutrons have no electrical charge.
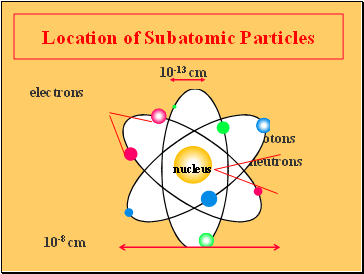
After the nucleus of the atom was discovered in 1911 by ernest rutherford, the nucleus of ordinary hydrogen was recognized to be a single proton. A negatively charged subatomic particle g. A positively charged subatomic particle 3. In 1932 the neutron was discovered. Typically in an atom, which two subatomic particles are equal in number? All matter in this universe is composed of atoms. The center of the atom is called the nucleus... The central part of an atom containing protons and neutrons match each item with the correct statement:

The center of the atom is called the nucleus.. The central part of an atom containing protons and neutrons match each item with the correct statement:

A negatively charged subatomic particle g. Atoms and molecules are called microscopic particles. A subatomic particle with no charge s. The central part of an atom containing protons and neutrons match each item with the correct statement: After the nucleus of the atom was discovered in 1911 by ernest rutherford, the nucleus of ordinary hydrogen was recognized to be a single proton. Protons have a positive (+) charge. Neutrons have no electrical charge. A positively charged subatomic particle 3. In particle physics, an elementary particle is a particle which cannot be split up into smaller pieces. Protons, neutrons, and electrons are the three main subatomic particles found in an atom.. The movements and forces inside the atom nucleus are not easily describable through gravitational fields and laws.

Subatomic particles are particles that are smaller than atoms.. Protons, neutrons, and electrons are the three main subatomic particles found in an atom. Atoms and molecules are called microscopic particles. Typically in an atom, which two subatomic particles are equal in number? Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. Neutrons have no electrical charge. A subatomic particle with no charge s. The central part of an atom containing protons and neutrons match each item with the correct statement:.. Subatomic particles are particles that are smaller than atoms.

The three main subatomic particles that form an atom are protons, neutrons, and electrons... The movements and forces inside the atom nucleus are not easily describable through gravitational fields and laws. Typically in an atom, which two subatomic particles are equal in number? He found out that there was no gravitational field in a, say, proton. Atoms and molecules are called microscopic particles. Subatomic particles include electrons, which are the negatively charged, almost massless particles which nevertheless account for most of the size of the atom. It includes the heavier building blocks of the small yet dense nucleus of the atom, the positively charged protons and the electrically neutral neutrons.. After the nucleus of the atom was discovered in 1911 by ernest rutherford, the nucleus of ordinary hydrogen was recognized to be a single proton.