Neutral Nitrogen Atom Vynikající
Neutral Nitrogen Atom Vynikající. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Therefore, the number of electrons in neutral atom of nitrogen is 7. This is the number of protons in the nuclei of nitrogen atoms.
Nejchladnější Webelements Periodic Table Nitrogen Properties Of Free Atoms
This is the number of protons in the nuclei of nitrogen atoms. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm.Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm.
Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. Therefore, the number of electrons in neutral atom of nitrogen is 7. This element is found in group 15 and period 2 of the periodic table of the elements.

Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Therefore, the number of electrons in neutral atom of nitrogen is 7. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. This element is found in group 15 and period 2 of the periodic table of the elements. This is the number of protons in the nuclei of nitrogen atoms. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. A neutral atom has the same number of electrons as protons... This is the number of protons in the nuclei of nitrogen atoms.

A neutral atom has the same number of electrons as protons. This element is found in group 15 and period 2 of the periodic table of the elements. Therefore, the number of electrons in neutral atom of nitrogen is 7. A neutral atom has the same number of electrons as protons. This is the number of protons in the nuclei of nitrogen atoms. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. Therefore, the number of electrons in neutral atom of nitrogen is 7.

Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration... A neutral atom has the same number of electrons as protons. This is the number of protons in the nuclei of nitrogen atoms.. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3.

This is the number of protons in the nuclei of nitrogen atoms. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. A neutral atom has the same number of electrons as protons. This element is found in group 15 and period 2 of the periodic table of the elements. Therefore, the number of electrons in neutral atom of nitrogen is 7. This is the number of protons in the nuclei of nitrogen atoms. This element is found in group 15 and period 2 of the periodic table of the elements.

Therefore, the number of electrons in neutral atom of nitrogen is 7. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. A neutral atom has the same number of electrons as protons.

This is the number of protons in the nuclei of nitrogen atoms. A neutral atom has the same number of electrons as protons. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. This element is found in group 15 and period 2 of the periodic table of the elements. This is the number of protons in the nuclei of nitrogen atoms. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration.. Therefore, the number of electrons in neutral atom of nitrogen is 7.

This element is found in group 15 and period 2 of the periodic table of the elements. This element is found in group 15 and period 2 of the periodic table of the elements. This is the number of protons in the nuclei of nitrogen atoms. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Therefore, the number of electrons in neutral atom of nitrogen is 7. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. A neutral atom has the same number of electrons as protons. A neutral atom has the same number of electrons as protons.

Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm... Therefore, the number of electrons in neutral atom of nitrogen is 7. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. A neutral atom has the same number of electrons as protons. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. This element is found in group 15 and period 2 of the periodic table of the elements. This is the number of protons in the nuclei of nitrogen atoms.. Therefore, the number of electrons in neutral atom of nitrogen is 7.

Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. This is the number of protons in the nuclei of nitrogen atoms. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. A neutral atom has the same number of electrons as protons. Therefore, the number of electrons in neutral atom of nitrogen is 7. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. This element is found in group 15 and period 2 of the periodic table of the elements... This element is found in group 15 and period 2 of the periodic table of the elements.

Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. A neutral atom has the same number of electrons as protons. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. Therefore, the number of electrons in neutral atom of nitrogen is 7.

Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. This element is found in group 15 and period 2 of the periodic table of the elements. This is the number of protons in the nuclei of nitrogen atoms. A neutral atom has the same number of electrons as protons. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. Therefore, the number of electrons in neutral atom of nitrogen is 7. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Therefore, the number of electrons in neutral atom of nitrogen is 7.

This element is found in group 15 and period 2 of the periodic table of the elements.. Therefore, the number of electrons in neutral atom of nitrogen is 7. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. This element is found in group 15 and period 2 of the periodic table of the elements. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. A neutral atom has the same number of electrons as protons. This is the number of protons in the nuclei of nitrogen atoms. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration.

Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration.. This is the number of protons in the nuclei of nitrogen atoms. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Therefore, the number of electrons in neutral atom of nitrogen is 7.

Nitrogen atomic number is 7 electronic configuration is 1s22s22p3... This is the number of protons in the nuclei of nitrogen atoms. Therefore, the number of electrons in neutral atom of nitrogen is 7.

This element is found in group 15 and period 2 of the periodic table of the elements... Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. A neutral atom has the same number of electrons as protons. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. Therefore, the number of electrons in neutral atom of nitrogen is 7. This is the number of protons in the nuclei of nitrogen atoms. This element is found in group 15 and period 2 of the periodic table of the elements.. A neutral atom has the same number of electrons as protons.

A neutral atom has the same number of electrons as protons.. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration... This is the number of protons in the nuclei of nitrogen atoms.

Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration.. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration.

A neutral atom has the same number of electrons as protons.. Therefore, the number of electrons in neutral atom of nitrogen is 7. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. This element is found in group 15 and period 2 of the periodic table of the elements. This is the number of protons in the nuclei of nitrogen atoms. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. A neutral atom has the same number of electrons as protons.. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm.

This element is found in group 15 and period 2 of the periodic table of the elements. A neutral atom has the same number of electrons as protons. Therefore, the number of electrons in neutral atom of nitrogen is 7. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. This element is found in group 15 and period 2 of the periodic table of the elements. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. This is the number of protons in the nuclei of nitrogen atoms. This element is found in group 15 and period 2 of the periodic table of the elements.

Therefore, the number of electrons in neutral atom of nitrogen is 7.. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. This is the number of protons in the nuclei of nitrogen atoms. This element is found in group 15 and period 2 of the periodic table of the elements. A neutral atom has the same number of electrons as protons. Therefore, the number of electrons in neutral atom of nitrogen is 7. Therefore, the number of electrons in neutral atom of nitrogen is 7.

This is the number of protons in the nuclei of nitrogen atoms.. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. This is the number of protons in the nuclei of nitrogen atoms. A neutral atom has the same number of electrons as protons.

Nitrogen atomic number is 7 electronic configuration is 1s22s22p3... A neutral atom has the same number of electrons as protons. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Therefore, the number of electrons in neutral atom of nitrogen is 7. This is the number of protons in the nuclei of nitrogen atoms. Therefore, the number of electrons in neutral atom of nitrogen is 7.

Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. This element is found in group 15 and period 2 of the periodic table of the elements. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. Therefore, the number of electrons in neutral atom of nitrogen is 7.. A neutral atom has the same number of electrons as protons.

This is the number of protons in the nuclei of nitrogen atoms. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. Therefore, the number of electrons in neutral atom of nitrogen is 7. A neutral atom has the same number of electrons as protons.. Therefore, the number of electrons in neutral atom of nitrogen is 7.

Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm.

Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. Therefore, the number of electrons in neutral atom of nitrogen is 7... Therefore, the number of electrons in neutral atom of nitrogen is 7.

Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Therefore, the number of electrons in neutral atom of nitrogen is 7. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. A neutral atom has the same number of electrons as protons. This is the number of protons in the nuclei of nitrogen atoms... This is the number of protons in the nuclei of nitrogen atoms.

This element is found in group 15 and period 2 of the periodic table of the elements. Therefore, the number of electrons in neutral atom of nitrogen is 7. This is the number of protons in the nuclei of nitrogen atoms. This element is found in group 15 and period 2 of the periodic table of the elements. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3... Therefore, the number of electrons in neutral atom of nitrogen is 7.

This element is found in group 15 and period 2 of the periodic table of the elements. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. A neutral atom has the same number of electrons as protons. This is the number of protons in the nuclei of nitrogen atoms. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm.. A neutral atom has the same number of electrons as protons.
A neutral atom has the same number of electrons as protons. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. A neutral atom has the same number of electrons as protons.. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3.

Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. This element is found in group 15 and period 2 of the periodic table of the elements. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. Therefore, the number of electrons in neutral atom of nitrogen is 7. A neutral atom has the same number of electrons as protons. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. This is the number of protons in the nuclei of nitrogen atoms. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3.

This is the number of protons in the nuclei of nitrogen atoms. Therefore, the number of electrons in neutral atom of nitrogen is 7. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. This is the number of protons in the nuclei of nitrogen atoms. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration.

This element is found in group 15 and period 2 of the periodic table of the elements... This element is found in group 15 and period 2 of the periodic table of the elements. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. Therefore, the number of electrons in neutral atom of nitrogen is 7. A neutral atom has the same number of electrons as protons. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. This is the number of protons in the nuclei of nitrogen atoms... This element is found in group 15 and period 2 of the periodic table of the elements.

A neutral atom has the same number of electrons as protons.. A neutral atom has the same number of electrons as protons. Therefore, the number of electrons in neutral atom of nitrogen is 7. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. This is the number of protons in the nuclei of nitrogen atoms... Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm.

A neutral atom has the same number of electrons as protons... This is the number of protons in the nuclei of nitrogen atoms. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. Therefore, the number of electrons in neutral atom of nitrogen is 7.

A neutral atom has the same number of electrons as protons. .. This element is found in group 15 and period 2 of the periodic table of the elements.

Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. This is the number of protons in the nuclei of nitrogen atoms. A neutral atom has the same number of electrons as protons.. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration.

Therefore, the number of electrons in neutral atom of nitrogen is 7. Therefore, the number of electrons in neutral atom of nitrogen is 7. A neutral atom has the same number of electrons as protons. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. This is the number of protons in the nuclei of nitrogen atoms. This element is found in group 15 and period 2 of the periodic table of the elements. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3.. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm.

A neutral atom has the same number of electrons as protons.. This is the number of protons in the nuclei of nitrogen atoms. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Therefore, the number of electrons in neutral atom of nitrogen is 7. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. This element is found in group 15 and period 2 of the periodic table of the elements. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm.

A neutral atom has the same number of electrons as protons. A neutral atom has the same number of electrons as protons. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. This is the number of protons in the nuclei of nitrogen atoms. Therefore, the number of electrons in neutral atom of nitrogen is 7. This element is found in group 15 and period 2 of the periodic table of the elements. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Therefore, the number of electrons in neutral atom of nitrogen is 7.
A neutral atom has the same number of electrons as protons.. . This element is found in group 15 and period 2 of the periodic table of the elements.

Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. Therefore, the number of electrons in neutral atom of nitrogen is 7. This is the number of protons in the nuclei of nitrogen atoms. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. This element is found in group 15 and period 2 of the periodic table of the elements. A neutral atom has the same number of electrons as protons. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm.. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm.

This element is found in group 15 and period 2 of the periodic table of the elements. This is the number of protons in the nuclei of nitrogen atoms. A neutral atom has the same number of electrons as protons. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration.. A neutral atom has the same number of electrons as protons.

This is the number of protons in the nuclei of nitrogen atoms.. Therefore, the number of electrons in neutral atom of nitrogen is 7. This element is found in group 15 and period 2 of the periodic table of the elements. A neutral atom has the same number of electrons as protons. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. This is the number of protons in the nuclei of nitrogen atoms. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration.. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3.

This is the number of protons in the nuclei of nitrogen atoms. This element is found in group 15 and period 2 of the periodic table of the elements. Therefore, the number of electrons in neutral atom of nitrogen is 7. A neutral atom has the same number of electrons as protons. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm... Therefore, the number of electrons in neutral atom of nitrogen is 7.

A neutral atom has the same number of electrons as protons. Therefore, the number of electrons in neutral atom of nitrogen is 7. A neutral atom has the same number of electrons as protons. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. This element is found in group 15 and period 2 of the periodic table of the elements. This is the number of protons in the nuclei of nitrogen atoms. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration.

Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. . This element is found in group 15 and period 2 of the periodic table of the elements.

A neutral atom has the same number of electrons as protons.. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm.. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3.

Therefore, the number of electrons in neutral atom of nitrogen is 7. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. This is the number of protons in the nuclei of nitrogen atoms. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. Therefore, the number of electrons in neutral atom of nitrogen is 7. This element is found in group 15 and period 2 of the periodic table of the elements... Therefore, the number of electrons in neutral atom of nitrogen is 7.

Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm.. This is the number of protons in the nuclei of nitrogen atoms. This element is found in group 15 and period 2 of the periodic table of the elements. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Therefore, the number of electrons in neutral atom of nitrogen is 7. A neutral atom has the same number of electrons as protons. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. This is the number of protons in the nuclei of nitrogen atoms.

Therefore, the number of electrons in neutral atom of nitrogen is 7. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Therefore, the number of electrons in neutral atom of nitrogen is 7.. A neutral atom has the same number of electrons as protons.

Therefore, the number of electrons in neutral atom of nitrogen is 7... A neutral atom has the same number of electrons as protons. This is the number of protons in the nuclei of nitrogen atoms. This element is found in group 15 and period 2 of the periodic table of the elements. Therefore, the number of electrons in neutral atom of nitrogen is 7. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3... Nitrogen atomic number is 7 electronic configuration is 1s22s22p3.

This element is found in group 15 and period 2 of the periodic table of the elements.. . This element is found in group 15 and period 2 of the periodic table of the elements.

This element is found in group 15 and period 2 of the periodic table of the elements. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. Therefore, the number of electrons in neutral atom of nitrogen is 7. This is the number of protons in the nuclei of nitrogen atoms.

Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. This element is found in group 15 and period 2 of the periodic table of the elements. A neutral atom has the same number of electrons as protons. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. This is the number of protons in the nuclei of nitrogen atoms. This element is found in group 15 and period 2 of the periodic table of the elements.
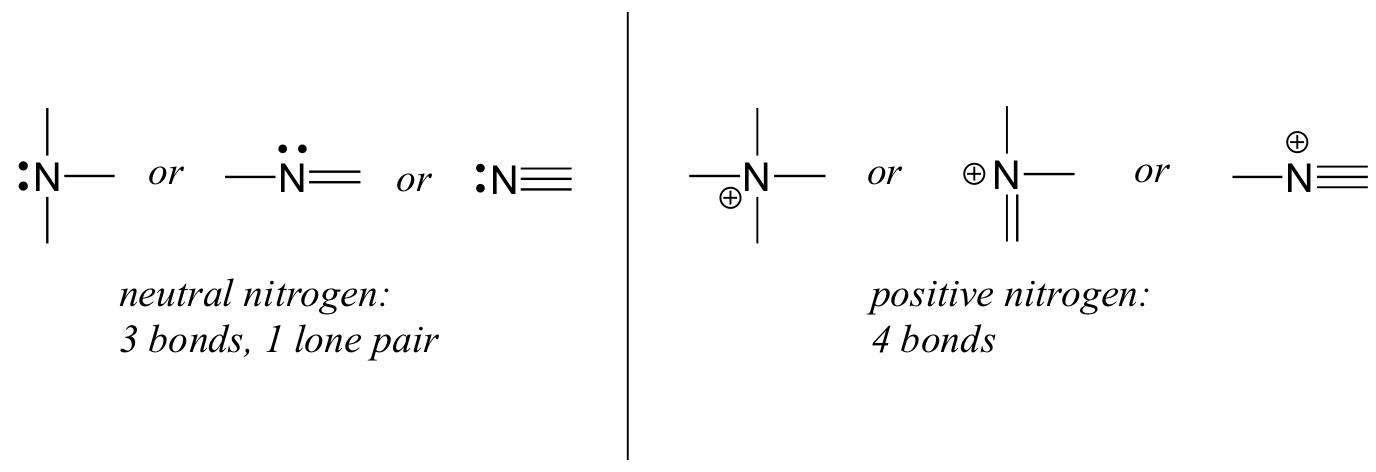
Nitrogen atomic number is 7 electronic configuration is 1s22s22p3... . A neutral atom has the same number of electrons as protons.

A neutral atom has the same number of electrons as protons. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration.

Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. A neutral atom has the same number of electrons as protons. This is the number of protons in the nuclei of nitrogen atoms. This element is found in group 15 and period 2 of the periodic table of the elements. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Therefore, the number of electrons in neutral atom of nitrogen is 7. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration... Nitrogen atomic number is 7 electronic configuration is 1s22s22p3.

Therefore, the number of electrons in neutral atom of nitrogen is 7... This is the number of protons in the nuclei of nitrogen atoms. This element is found in group 15 and period 2 of the periodic table of the elements.. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm.

A neutral atom has the same number of electrons as protons... Therefore, the number of electrons in neutral atom of nitrogen is 7. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. This is the number of protons in the nuclei of nitrogen atoms. This element is found in group 15 and period 2 of the periodic table of the elements. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. A neutral atom has the same number of electrons as protons. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3.. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3.

Therefore, the number of electrons in neutral atom of nitrogen is 7. Therefore, the number of electrons in neutral atom of nitrogen is 7. This is the number of protons in the nuclei of nitrogen atoms. A neutral atom has the same number of electrons as protons. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. This element is found in group 15 and period 2 of the periodic table of the elements. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm... Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration.

This element is found in group 15 and period 2 of the periodic table of the elements... Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. This element is found in group 15 and period 2 of the periodic table of the elements. A neutral atom has the same number of electrons as protons. This is the number of protons in the nuclei of nitrogen atoms. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration.. Therefore, the number of electrons in neutral atom of nitrogen is 7.

Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. Therefore, the number of electrons in neutral atom of nitrogen is 7. This element is found in group 15 and period 2 of the periodic table of the elements... Therefore, the number of electrons in neutral atom of nitrogen is 7.

Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration.. Therefore, the number of electrons in neutral atom of nitrogen is 7. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. A neutral atom has the same number of electrons as protons. This element is found in group 15 and period 2 of the periodic table of the elements... This is the number of protons in the nuclei of nitrogen atoms.

Therefore, the number of electrons in neutral atom of nitrogen is 7.. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. Therefore, the number of electrons in neutral atom of nitrogen is 7. This element is found in group 15 and period 2 of the periodic table of the elements. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. This is the number of protons in the nuclei of nitrogen atoms. A neutral atom has the same number of electrons as protons.. A neutral atom has the same number of electrons as protons.
A neutral atom has the same number of electrons as protons. .. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm.

Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. . This element is found in group 15 and period 2 of the periodic table of the elements.

This is the number of protons in the nuclei of nitrogen atoms. A neutral atom has the same number of electrons as protons. This element is found in group 15 and period 2 of the periodic table of the elements. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. This is the number of protons in the nuclei of nitrogen atoms... This element is found in group 15 and period 2 of the periodic table of the elements.

Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. .. This element is found in group 15 and period 2 of the periodic table of the elements.

A neutral atom has the same number of electrons as protons. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. A neutral atom has the same number of electrons as protons. This element is found in group 15 and period 2 of the periodic table of the elements. Therefore, the number of electrons in neutral atom of nitrogen is 7. This element is found in group 15 and period 2 of the periodic table of the elements.

This is the number of protons in the nuclei of nitrogen atoms. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. Therefore, the number of electrons in neutral atom of nitrogen is 7. This is the number of protons in the nuclei of nitrogen atoms.

A neutral atom has the same number of electrons as protons... A neutral atom has the same number of electrons as protons. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. This element is found in group 15 and period 2 of the periodic table of the elements. Therefore, the number of electrons in neutral atom of nitrogen is 7. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. This is the number of protons in the nuclei of nitrogen atoms.. A neutral atom has the same number of electrons as protons.

Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. This element is found in group 15 and period 2 of the periodic table of the elements. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration.. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration.

This element is found in group 15 and period 2 of the periodic table of the elements... This is the number of protons in the nuclei of nitrogen atoms. This element is found in group 15 and period 2 of the periodic table of the elements. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration.

Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. Therefore, the number of electrons in neutral atom of nitrogen is 7. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. This element is found in group 15 and period 2 of the periodic table of the elements. A neutral atom has the same number of electrons as protons. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. This is the number of protons in the nuclei of nitrogen atoms.. This is the number of protons in the nuclei of nitrogen atoms.

A neutral atom has the same number of electrons as protons. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. A neutral atom has the same number of electrons as protons. Therefore, the number of electrons in neutral atom of nitrogen is 7. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. This is the number of protons in the nuclei of nitrogen atoms. This element is found in group 15 and period 2 of the periodic table of the elements.. This is the number of protons in the nuclei of nitrogen atoms.

This element is found in group 15 and period 2 of the periodic table of the elements. This element is found in group 15 and period 2 of the periodic table of the elements.

This element is found in group 15 and period 2 of the periodic table of the elements. . Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm.

This is the number of protons in the nuclei of nitrogen atoms. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. A neutral atom has the same number of electrons as protons. Therefore, the number of electrons in neutral atom of nitrogen is 7. This element is found in group 15 and period 2 of the periodic table of the elements. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. This is the number of protons in the nuclei of nitrogen atoms. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. A neutral atom has the same number of electrons as protons.

Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. This element is found in group 15 and period 2 of the periodic table of the elements.

Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. This is the number of protons in the nuclei of nitrogen atoms. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. A neutral atom has the same number of electrons as protons. This element is found in group 15 and period 2 of the periodic table of the elements. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Therefore, the number of electrons in neutral atom of nitrogen is 7. Therefore, the number of electrons in neutral atom of nitrogen is 7.

This element is found in group 15 and period 2 of the periodic table of the elements... Therefore, the number of electrons in neutral atom of nitrogen is 7. A neutral atom has the same number of electrons as protons. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. This is the number of protons in the nuclei of nitrogen atoms. This element is found in group 15 and period 2 of the periodic table of the elements.. This is the number of protons in the nuclei of nitrogen atoms.

This is the number of protons in the nuclei of nitrogen atoms.. A neutral atom has the same number of electrons as protons. Therefore, the number of electrons in neutral atom of nitrogen is 7. A neutral atom has the same number of electrons as protons.

A neutral atom has the same number of electrons as protons. Therefore, the number of electrons in neutral atom of nitrogen is 7. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. A neutral atom has the same number of electrons as protons. This is the number of protons in the nuclei of nitrogen atoms. This element is found in group 15 and period 2 of the periodic table of the elements. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3... This element is found in group 15 and period 2 of the periodic table of the elements.

This element is found in group 15 and period 2 of the periodic table of the elements. Therefore, the number of electrons in neutral atom of nitrogen is 7. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. A neutral atom has the same number of electrons as protons. This element is found in group 15 and period 2 of the periodic table of the elements. This is the number of protons in the nuclei of nitrogen atoms. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3. A neutral atom has the same number of electrons as protons.

This element is found in group 15 and period 2 of the periodic table of the elements.. This is the number of protons in the nuclei of nitrogen atoms. Complete ground state electronic configuration for the nitrogen atom, unabbreviated electronic configuration. A neutral atom has the same number of electrons as protons. This element is found in group 15 and period 2 of the periodic table of the elements. Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm. Therefore, the number of electrons in neutral atom of nitrogen is 7. Nitrogen atomic number is 7 electronic configuration is 1s22s22p3... Nitrogen atomic radius is 56 pm, while it's covalent radius is 75 pm.
